Seeing and Engaging the Light
INTRODUCTION
The expected high ranking of lasers, unique in class technology, merits attention to understand why they lack clinical market penetration in dentistry. Their validated potential and similar target market position have already been captured for decades in other healthcare sectors. I will discuss in broad strokes the top 3 currently relevant self-imposed limitations in utilizing lasers in dentistry. These are (1) incomplete and incorrect optical physics education (excellent clinicians practice science), (2) magnification (laser parameters demand magnification), and (3) inadequate device packaging of wavelength and parameter selection as well as coordination (medical device technology packaging). Three solutions will be presented to these historical reflections. Modification of these 3 variables in function of not only the present, but future considerations for integration, is essential in transforming pathology to healthy tissue. No other technology does it like lasers.
Optical physics technology has been readily available in the medical sector, successfully applied, and integrated into cutting-edge clinical outcome techniques, with more temperamental pathologic characterization as compared to simple pathology. Although it contributed to the first springboard of laser applications to tissue, the dental sector had the opportune time to bring this technology to the forefront and seemingly did not. We have seen a great concern for its minimal utilization compared to its proven potential, even in difficult clinical scenarios. It has lagged in market penetration, and the question is why?
EDUCATION
It is unfortunate that with increased resources, more data, and magnified research efforts, the field of lasers in dentistry has suffered from little penetration of accurate laser physics in its educational and training spaces. A precise understanding of physics, light-transfer biomaterials, and interaction with tissue are determinants of clinical application modification and application in reproducible, optimal outcomes. It is not a mere race from point A to point B. The terrain that stands between the points requires the technology selection and renders its superlative clinical outcome. In this sense, it is not like any other technology in medical devices. Optical and quantum physics-based medical devices generally fall into 2 categories: diagnostic and intervention. Both require real-time modification in function of subject tissue profiles, with the latter often requiring the most customization in the dental and oral science sector. Homogeneity, by virtue of generalizations of historical grouping of clinical phenotypes, is a luxury of the past. The best clinicians practice science in real time. Clinicians are scientists. Case in point, the “do no harm and do all to heal” clause of the Hippocratic Oath requires that very protocol customization and optimization rendered by credentials, experience, and knowledge.
The solution: Accurate and comprehensive optical physics education is the dependent variable for device scrutiny and selection.
MAGNIFICATION
Laser capability and function, by definition, rests within such small parameters as only magnification can appropriately idealize during reading and execution. Few diagnostic technology packages have made it to the dental market, although the demand is great and the medical market already enjoys a wide historical range of applications of that caliber. For the intervention aspect of laser technology, magnification is the most appropriate match of delivery to really enhance access to the applications of laser technology. Tissue differentiation claims different laser setting potential and can and will result in a high precision of clinical outcome vs a typical conventional outcome. A great example of this is depicted in the optimization of tissue treatment without loss of tissue volume, as one clinical case below will showcase. Without magnification and specified tissue geo-position, the laser technology can be easily underutilized.
The solution: The incorporation of segmental range of visualization selection in function of pathology characterization is key to laser settings and modality of application. Stop points and laser settings become crucial for optimal results, while magnification guides and ensures this.
TECHNOLOGY PACKAGING
The expanded medical sector has enjoyed continuous application of optical physics from neurology to dermatology, ophthalmology, and cardiovascular disciplines, to name a few. Dentistry, though it encases a smaller volume extension per body size, corresponds to extremely high-differentiated tissue subtypes in extremely close proximity. And yet, the laser technology packaging has remained poor and undifferentiated compared to that of other disciplines. Medical device packaging includes the package insert, which stipulates various elements that comprise medical device developments, claims, parameters, and technical features. The existing available collection of laser equipment in dentistry could easily improve in quality, quantity, and variation of referenced technology with very little cost efforts from manufacturing to market launch, while the cost-benefit analysis is favorable for all parties involved. The resulting outcome translates in generating excellent clinical results with minimal invasion; less instrumentation requirements, including anesthesia; and improved healing potential, which supersedes the conventional sequence manifestation and inadvertently should translate into lower cost. However, incomplete or expensive technology packaging choices can hurt access to superior treatment options in certain pathologies in the long run, which can even eliminate the product’s availability and participation in the marketplace. Logic in medical device development can make or break a clinical protocol even if demand is high, resulting in generating clinical phenotypes, which can perpetuate higher prevalence and incidence rates with increased proliferation of additional clinical demands. For example, choice in wavelength utilization in oral viral lesions can and will result in different profiles of clinical outcomes and recurrence. Laser profiles are largely defined by type of wavelength and other parameters, which make lasers devices that are sensitive in selection to precision, and hence, industry participation needs to parallel research and development support.
The solution: The incorporation of segmental range of visualization in function of pathology characterization. This is the key to laser choice, settings, and modality of application—ie, hot tip technology does not have the same impact as laser optical technology and its associated parameters.
CLINICAL ANGLE
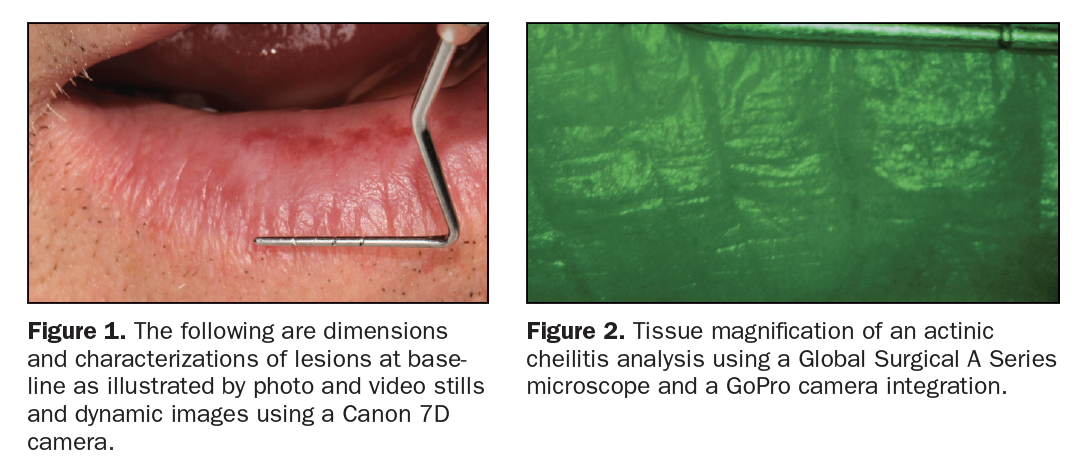
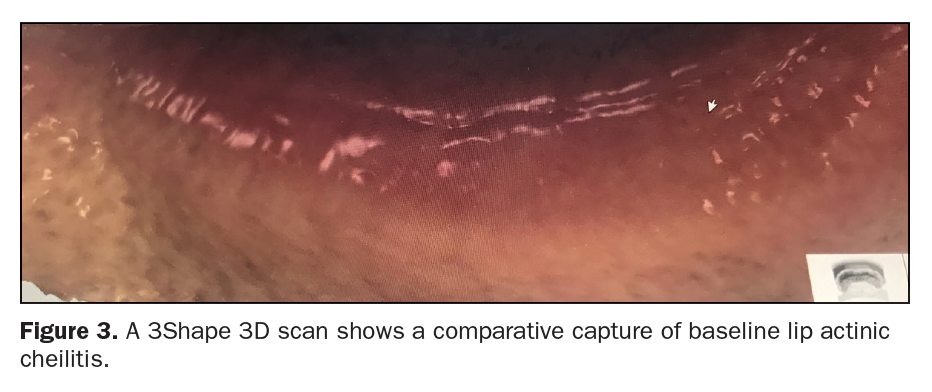
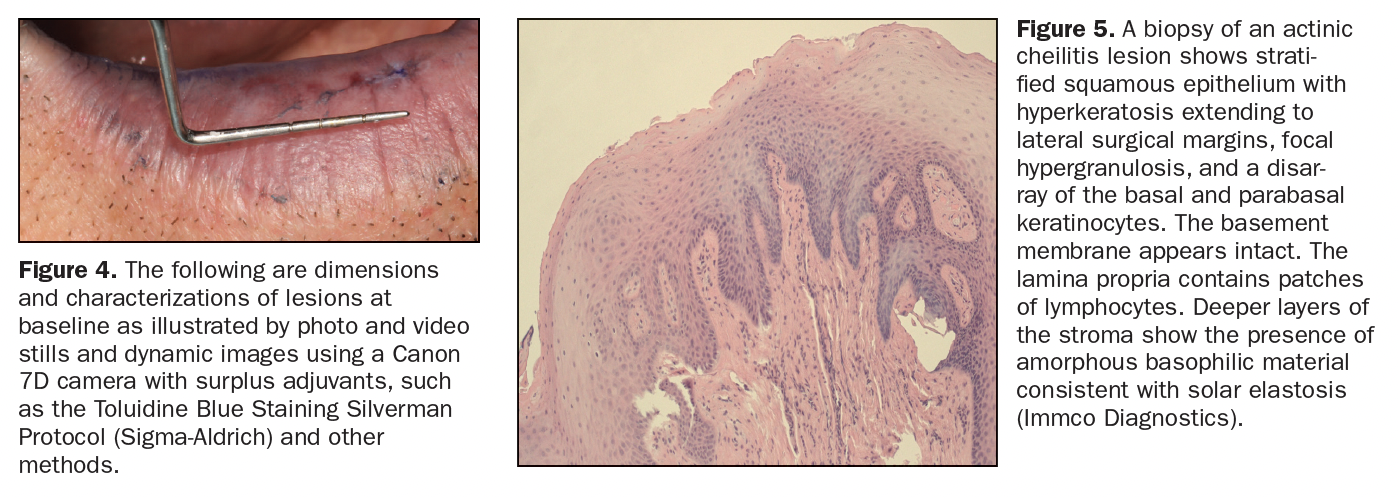
Examples of clinical cases outlined may separately exemplify similar limited portions of a larger and complete full protocol, pending publication. The following cases and illustrations will demonstrate the various adaptations of the above discussion and premise. The dimension and characterization of lesions at the baseline are illustrated by photo and video stills and dynamic images with a Canon 7D Camera (Figure 1), A Series Microscope (Global Surgical) (Figure 2), 3D Scanner (3Shape) (Figure 3), and with surplus adjuvants like the Toluidine Blue Staining Silverman Protocol (Figure 4)1 and other methods.
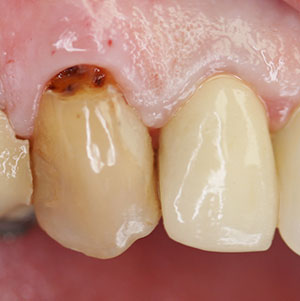
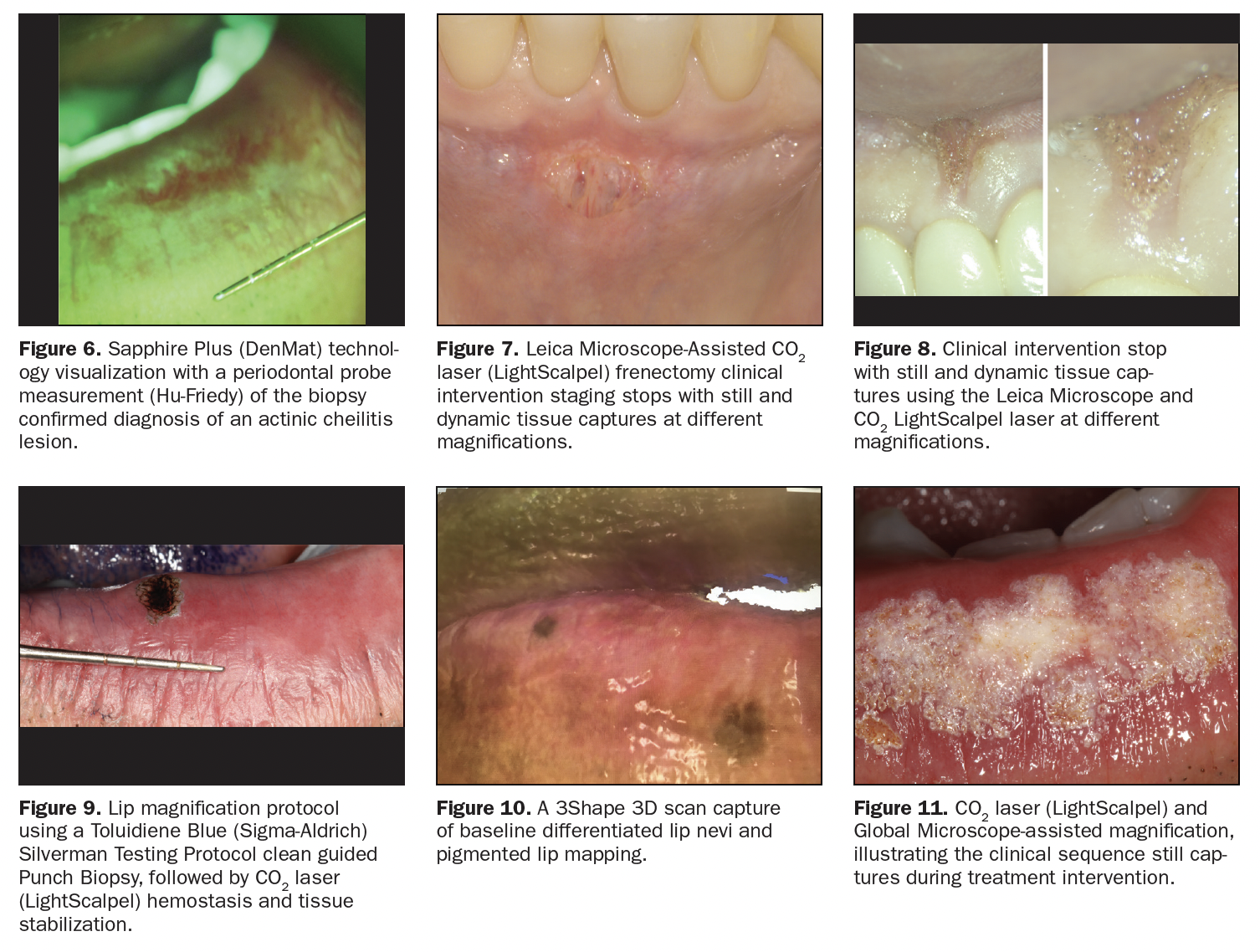
Figure 5 shows a baseline lip diagnosis of a 56-year-old male ASA I illustrating actinic cheilitis by histopathology biopsy (IMMCO Diagnostics) in various portions of the lip extending from the vermilion border. Lesion dimension, measurements, and real-time characterization of tissue can be obtained with the utilization of a probe instrument (Hu-Friedy Instruments) and other methods, including using the Sapphire Plus (DenMat), as shown in Figure 6. To optimize comprehensive treatment with a low risk of recurrence and minimal or no volumetric loss, a selection of wavelength and parameters finalized the choice to the CO2 wavelength (LightScalpel) and set parameters with and without the utilization of topical anesthetic (4% Oroquix), depending on patient request, lesion position, and tissue depth. The CO2 choice of wavelength in treating actinic cheilitis has been reported as a leading choice with optimal results and a low risk of recurrence.2 Further customization to eliminate the risk of volumetric loss of tissue is especially relevant in moderate to large lesions, and early intervention opportunities are necessary. The full protocol is pending publication (Washington Institute for Dentistry & Laser Surgery).
Figure 7, a different case, shows tissue under magnification (Leica Microscope) during the intervention phase of a frenectomy, which precisely determined the optimization of speed and stop points utilizing the CO2 laser wavelength (LightScalpel) vs other lasers or packages, such as hot tip light transfer technologies. Figures 3, 8, 9, 10 and 11 also illustrate various views of baseline to clinical intervention tissue stops with still and dynamic tissue captures using an A Series Microscope, a Toluidine Blue (Sigma-Aldrich) Testing Protocol Silverman guided Punch Biopsy (C3 Think Tank, Washington Institute for Dentistry & Laser Surgery), and an Apple iPhone capture of a 3Shape scan. These varied references have clearly made the case for supporting the points presented in the scope of this article.
CONCLUSION
In summary, self-limiting factors have brought dentistry considerable consequences and setbacks in laser-technology adoption. The scope of translational research should make it easier to adapt cross-discipline discoveries and allow for quick cross-market launch. However, the core of laser technology cannot be ignored nor forgotten as adaption occurs, while prototypical product development strategies cannot override and ultimately sacrifice its original technological core potential. Optical physics education is crucial in tissue matching and recognition of pathological progression and when making the choice of a laser and its inherent required parameters and settings, which more often than not should always be customized. Visualization to match laser-technology potential is a necessary consideration to enjoy a full range of capabilities in clinical outcome optimization in interdisciplinary dentistry.
ACKNOWLEDGEMENT
All images and content/concepts in this article are proprietary of C3 Think Tank, Washington Institute for Dentistry & Laser Surgery.
REFERENCES
1. Silverman S Jr, Migliorati C, Barbosa J. Toluidine blue staining in the detection of oral precancerous and malignant lesions. Oral Surg Oral Med Oral Pathol. 1984;57(4):379–82. doi:10.1016/0030-4220(84)90154-3
2. Shah AY, Doherty SD, Rosen T. Actinic cheilitis: a treatment review. Int J Dermatol. 2010;49(11):1225–34. doi:10.1111/j.1365-4632.2010.04580.x
ABOUT THE AUTHOR
Dr. Cotca received a Bachelor of Science degree in Chemistry and Cellular Molecular Biology, a Master’s degree in Public Health and Toxicology, and a DDS degree from University of Michigan. She is an international lecturer and aesthetic restorative dentist and founded the Washington Institute for Dentistry & Laser Surgery in the Washington, DC, area—a private practice institute, a C3 Think Tank, and an innovative, real-time protocol incubator where she develops advanced oral systemic aesthetic clinical protocols. Dr. Cotca consults in medical device and clinical protocol development and serves on the ADA Standards Committee on Dental Products and as the US Delegate to the International Organizations of Standardization, among many others.
She serves as an editor and reviewer of various peer-reviewed oral sciences journals and is a Fellow of the American Academy of Oral Medicine, the Pierre Fauchard Academy, and the International College of Dentists. Dr. Cotca is also a member of the International College of Prosthodontists. She has been an active expert on Capitol Hill, testifying before the United States Congress and the White House, and she is a dental expert and contributor for ABC and NBC and in the healthcare industry sectors. She can be reached at claudiaccotca.com.
Disclosure: Dr. Cotca reports no disclosures.
RELATED ARTICLES
Focus On: Laser Dentistry for the General Practice
Practical Laser Applications in General Practice
Dental Lasers and Peri-Implantitis – 5 Questions With Dr. Samuel Low