Dental patients taking anticoagulant medication pose a challenge for the clinician. Dentists are often required to manage bleeding as part of routine oral surgery or dental procedures, and altered hemostasis can lead to complications. Nevertheless, use of these medications is generally important for the patientís health, and any alterations in the anticoagulant regimen may have untoward sequelae. In addition, several medications can affect the clotting mechanism, potentially compromising hemostasis. This article will review a variety of anticoagulant medications and the medical conditions that necessitate their use. In addition, recommendations for altering anticoagulant medication prior to performing routine oral surgery will be reviewed.
 |
|
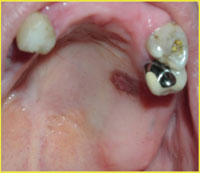
Figure 1. A patient with vascular fragility due to long-term corticosteroid use presented with this localized bruise as a result of irritation from an ill-fitting partial denture. |
Hemostasis depends on several critical factors.1 An adequate number of platelets and proper platelet function are essential. The integrity of the vasculature also plays an important role in hemostasis. Vascular integrity can be compromised by vitamin C deficiency, viral and bacterial infections, or as a result of the aging process or through various disease states (Figure 1). Adequate levels of clotting factors and proper functioning of the fibrinolytic pathway are also essential. Finally, many pharmaceuticals can compromise hemostasis, which is the focus of this article. Chemically induced anticoagulation as a therapeutic strategy is required treatment for a variety of diseases. Anticoagulation is also an undesirable side effect of some drugs.1
Many drugs can interfere with hemostasis. In addition to anticoagulant medications, these agents include antibiotics, aspirin, and anti-inflammatory medications. Drugs used in the treatment of cancer can interfere with the clotting mechanism, as does excessive alcohol intake. The dentist should be aware of any herbal supplements the patient is taking, as a number of these can also affect hemostasis.1
LABORATORY TESTS
The coagulation cascade involves the intrinsic, extrinsic, and common pathways, which have been found to be highly interconnected. The intrinsic pathway requires the clotting factors VII, IX, X, XI, and XII. The extrinsic pathway is initiated with the release of tissue factor II. The common point in both pathways is the activation of factor X to factor Xa. Factor Xa activates prothrombin (factor II) to thrombin (factor IIa). Thrombin, in turn, converts fibrinogen to fibrin.
Dentists should be able to order and interpret appropriate laboratory tests when minor oral surgery is required for anticoagulated patients. The most frequently used tests are the Prothrombin Time (PT) and International Normalized Ratio (INR).
The PT measures the effectiveness of the extrinsic and common pathways. The normal value is approximately 10 to 15 seconds. Because of the variability in PT reported by different laboratories, it is no longer considered adequate to use PT to monitor the level of anticoagulation. In order to reduce variability, INR was developed as a consistent test for measuring anticoagulation status. INR is the more reliable and consistent measure. A normal individual should have an INR of 1.0.2
Dependent upon the medical condition, physicians will prescribe anticoagulant medication so that patients demonstrate an INR in a therapeutic range of approximately 2.0 to 3.5. These conditions include states in which thromboembolism could cause serious morbidity or death, such as post myocardial infarction, and prevention of deep venous thrombosis (DVT). Additionally, patients with atrial fibrillation are anticoagulated because of the greater likelihood of blood clot formation due to turbulent blood flow through the heart. In those individuals with prosthetic heart valves and certain hypercoagulable states, the INR should be approximately 3.5. If the dentist encounters a patient with an INR greater than 4.0, the patient should be referred to the physician for evaluation. An INR greater than 4.0 is usually considered nontherapeutic, and the patient is at risk for serious bleeding complications.3
Another important test used to assess coagulation is the activated Partial Thromboplastin Time (aPTT), which measures the intrinsic and common pathways. A normal test time is 25 to 40 seconds. The aPTT is commonly used to measure the effectiveness of the anticoagulant heparin. This medication is typically administered intravenously on an inpatient basis.1 Heparin therapy will be discussed in more detail later in this article.
A clinical laboratory test that may be used to monitor platelet function is Bleeding Time. Bleeding Time is measured by making a small incision on the forearm while a blood pressure cuff, inflated to 40 mm Hg, is placed on the upper arm. The time for the bleeding to stop is measured. Normal Bleeding Time is in the range of 2 to 8 minutes. This test can be quite variable and is generally used as a screening tool or in combination with other tests.1
As opposed to the Bleeding Time, a platelet count is more likely to be available in the patient’s medical record. Normal platelet counts are in the range of 100,000 to 400,000/mm3. If the count falls below 100,000/mm3, then the patient is considered to have thrombocytopenia. Thrombocytopenia represents a potential for compromised hemostasis. When the platelet count falls below 50,000/mm3, the risk of prolonged bleeding is especially significant. Spontaneous hemorrhage may occur when the platelet count falls below 25,000/mm3. Thrombocytopenia is frequently associated with a serious medical condition. It can be drug-induced, caused by bone marrow failure, hypersplenism, or cancer chemotherapy, or be of idiopathic origin.1
|
Table 1. Herbal Supplements That Affect Platelet Aggregation: Agents and Uses. |
|
Garlic – lowers cholesterol and blood pressure |
A situation more commonly encountered in the dental office is thrombocytopathy. This condition represents a state of compromised platelet function and aggregation. Thrombocytopathy can be associated with aspirin, nonsteroidal anti-in-flammatory drugs (NSAIDs), and drugs used to decrease the risk for stroke or heart attack, such as Aggrenox (aspirin/extended-release dipyridamole; Boehringer Ingelheim Pharmaceuticals) and Plavix (clopidogrel bisulfate; Bristol-Myers Squibb/ Sanofi Pharmaceuticals Partnership). Certain herbal supplements can be associated with compromised platelet aggregation (Table 1). It is recommended that patients who are scheduled for oral surgery refrain from taking any herbal substances for 1 week prior to the surgery.4,5
ANTICOAGULANT MEDICATIONS
Patients seen in the dental office may have been instructed by their physician to take one low-dose aspirin daily to prevent thromboembolic events associated with stroke or myocardial infarction. One low-dose aspirin contains 81 mg, whereas one regular-strength aspirin contains 325 mg. Some health professionals recommend taking 325 mg every other day. Cessation of low-dose aspirin therapy prior to oral surgery has been controversial. Dentists have been concerned with potential bleeding complications; however, patients who are receiving aspirin therapy are at risk for emboli and myocardial infarction if the medication is stopped.6 Studies have shown that patients undergoing minor oral surgery, including implant surgery and third molar extraction, experienced minimal bleeding complications when on low-dose aspirin therapy. These complications were controlled with local measures. The results indicated that low-dose aspirin therapy may be continued even if a patient requires minor oral surgery.7,8
Medications such as Plavix or Aggrenox are prescribed to prevent stroke and heart attack. Plavix irreversibly binds to the platelets, and Aggrenox combines 25 mg of aspirin and 200 mg of extended-release dipyridamole, providing additive antiplatelet actions. Dipyrid-amole reversibly inhibits adenosine uptake leading to increased cyclic AMP, and inhibits cyclic GMP phosphodiesterase resulting in an increase of cyclic GMP. Both products are platelet antiaggregants. These medications may also be continued during minor oral surgical procedures. Local measures should be sufficient to control bleeding.9 If any significant bleeding is anticipated, dentists should consult with the patientís physician to determine if alterations to the anticoagulant regimen should be considered.
Another class of drug that disrupts platelet activity is the NSAIDs. These medications reversibly bind to the platelet for a limited period of time (approximately 6 hours). Used by themselves, there should be no complications if a patient is taking a NSAID. Complications can arise if the patient is taking anticoagulant medication and a NSAID. This combination could increase the risk for bleeding.10
|
Table 2. Target INR for Specific Medical Conditions. |
||||||||||||
|
The medication of greatest concern relative to bleeding complications, and that is a source of confusion for many practicing dentists, is the anticoagulant warfarin sodium, which is often referred to by the brand name Coumadin (Bristol-Myers Squibb Company).11 Coumadin is used to prevent thromboembolic events that can occur in a variety of conditions. These include ischemic heart disease, deep venous thrombosis and pulmonary embolism, the presence of artificial heart valves, and hypercoagulable disease states. Genetic or acquired diseases include antiphospholipid anti-bodies (Lupus Anticoagulant), deficiencies of antithrombin, protein C and its co-factor protein S, and Factor V Leiden. It should be noted that most patients with Lupus Antico-agulant do not have lupus erythematosus or other systemic autoimmune disorders. Factor V Leiden is an autosomal dominant condition in which Factor V remains active, which facilitates overproduction of thrombin, leading to excess fibrin generation and excess clotting. It is named after the city of Leiden (Netherlands), where it was first identified in 1994. The effect of Coumadin is measured by the INR or PT.1 Typical INR values for specific medical conditions requiring Coumadin are listed in Table 2.
Coumadin has a mean half-life of 40 hours, which can vary among individuals from 20 to 60 hours (Physician’s Desk Reference). Coumadin interferes with the synthesis of vitamin K. Vitamin K is a necessary co-factor in chemical reactions at numerous points in the coagulation cascade. Numerous drug interactions are possible with this medication. The mechanism may be pharmacokinetic, in that the absorption, protein binding, or hepatic metabolism of warfarin is affected. In addition, drugs that affect vitamin K production can also alter hemostasis. Of particular importance to dentists are broad-spectrum antibiotics. These drugs can cause changes in the intestinal flora that might decrease vitamin K absorption.12,13 One should be aware that multiple tooth extractions may make eating difficult and enhance the antibiotic-induced decrease in vitamin K availability. If it is necessary to prescribe a course of antibiotics, then the dentist may wish to consider having the patientís INR monitored. There are case reports demonstrating this interaction with single-dose amoxicillin prophylaxis recommended by the American Heart Association. There is no evidence to suggest that single-dose clindamycin interacts with warfarin.14
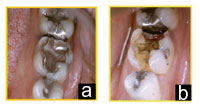 |
| Figures 2a and 2b. Altering the dosage of anticoagulant medication would not be necessary when replacing this defective amalgam restoration. |
Until recently, many dentists and physicians were of the belief that Coumadin should be discontinued in low-risk to moderate-risk patients undergoing dental procedures associated with bleeding. Some dentists even requested that the patient stop taking Coumadin for restorative or nonsurgical periodontal procedures (Figures 2a and 2b). However, there are documented cases of embolic complications in patients whose Coumadin therapy was discontinued for dental treatment.15,16 In addition, there is evidence that thrombosis may also occur because of a temporary state of rebound hypercoagulability following cessation of anticoagulation therapy.17
Traditionally, when a patient at high risk for thrombus formation required oral surgey, hospitalization was required. High-risk patients include individuals with mechanical heart valves, those undergoing active treatment for DVT, or those with hypercoagulable disease states.18 These patients had their Coumadin discontinued and were placed on intravenous heparin. Although this remains the case for major oral surgery procedures, this approach was also used for relatively minor surgical procedures. Heparin is a mixture of glycosaminoglycans of different molecular weights. Heparin potentiates the action of antithrombin III and thereby inactivates thrombin (factor IIa) as well as factors IX, X, XI, XII, and plasmin, and prevents the conversion of fibrinogen to fibrin. This unfractionated heparin was generally administered intravenously in the hospital. It has a short half- life of about 50 to 90 minutes. When a surgical procedure was planned, the heparin was discontinued 4 to 6 hours prior to the procedure and restarted 4 to 6 hours later.19,20 As a result, the period of time a patient was not anticoagulated was only a few hours. As the Coumadin the patient had been taking was metabolized and eliminated from the body, heparin remained as the anticoagulant. Two or three days after dental surgery was performed, the patient was restarted on Coumadin. The Coumadin was allowed to reach therapeutic levels, and the heparin was discontinued. This process required approximately 5 days of hospitalization and close patient monitoring.
In the past several years, low molecular weight heparins (LMWHs) have been introduced. This relatively new class of agents is ob-tained from chemical or enzymatic depolymerization of unfractionated heparin. Low molecular weight heparins have been used to prevent deep venous thrombosis and pulmonary emboli. In addition, they are used in the treatment of unstable angina and non-Q wave myocardial infarcts, and for embolism prophylaxis following major orthopedic and abdominal surgery.19,20 Low molecular weight heparins have more recently been used as bridging therapy in order to discontinue Coumadin in preparation for dental procedures. Low molecular weight heparins are not approved as a substitute for Coumadin in patients with prosthetic heart valves.20,21 Patients can self-administer these drugs subcutaneously on an outpatient basis. As an alternative to the previous technique involving unfractionated heparin, low molecular weight heparins have provided successful anticoagulation, require less monitoring, reduce the duration and cost of hospital admissions, and provide the convenience for dental procedures, which can be completed entirely on an outpatient basis.
Although there are isolated cases of postextraction hemorrhage, many of these cases involved full-mouth extractions and alveoplasties, and some of the patientsí anticoagulation levels exceeded therapeutic levels.22 When following current recommendations, only about 1% of patients experienced a significant postoperative bleeding episode.18,22,23 All of these episodes were controllable with local measures. Never-theless, the level of anticoagulation, risk of bleeding from the procedure, risk of thromboembolism, and the patientís overall systemic health must be considered. For example, patients with a mechanical heart valve, atrial fibrillation with a history of stroke, and individuals with hypercoagulable disease states are at high risk for thromboembolism.
|
Table 3. Clinical Trials Studying Continuous Anticoagulation and Dentoalveolar Surgery Summarized by Beirne.18 |
||||||||||||||||||||||||||||
|
Numerous investigators have shown that modification of anticoagulant medication is not necessary when performing routine dentoalveolar surgery. An article by Beirne18 summarizes these clinical studies (Table 3). The article concludes that, “Stopping warfarin with or without bridging is not supported by clinical evidence. The risk of developing life-threatening bleeding that cannot be controlled using local measures following dental extractions, alveoplasties, or dental implants is so low that there is no need to stop warfarin.”
MAINTAINING HEMOSTASIS
|
Table 4. Commercial Agents Available to Aid in Control of Bleeding. |
|
Gelfoam (Pfizer) |
With the recommendation not to alter anticoagulant therapy for patients requiring routine dentoalveolar surgery, dentists must adhere to meticulous surgical technique, have the skills to ensure proper wound closure, and be familiar with the adjunctive hemostatic techniques (Table 4). Prior to performing surgery, it is recommended that the dentist obtain the appropriate laboratory values that assess coagulation.24
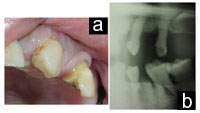 |
| Figures 3a and 3b. Pretreatment status of a patient taking Coumadin prior to extraction of these 2 maxillary teeth. |
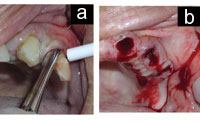 |
| Figures 4a and 4b. Extraction and immediately after extraction, anticoagulated condition can be appreciated in Figure 4b. |
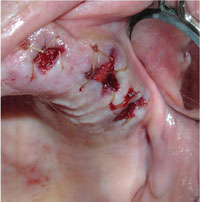 |
| Figures 5. Excellent hemostasis is achieved with local measures. (Same patient as in Figure 4.) |
For the anticoagulated patient, applying pressure is still an excellent initial step in obtaining hemostasis. This approach will probably not be sufficient by itself. Even if hemostasis is obtained in the dental office, the surgical site is likely to be disturbed as the patient eats or speaks, thus requiring an after-hours appointment to re-establish hemostasis. At a minimum the dentist should consider using a resorbable gelatin sponge (Gelfoam [Pfizer]) inserted into the extraction socket prior to suturing (Figures 3 through 5). Gelfoam acts as a mechanical matrix to facilitate clotting. Resorbable oxidized cellulose (Surgicel [Johnson & Johnson]) is also well-known to dentists and is used similarly to Gelfoam. It should be noted that any type of packing material can be a nidus for infection, and caution in the use of these products should be the rule when operating in an infected area.25
Hemostatic collagen (Colla-Plug [Integra LifeSciences]) provides a mechanical ma-trix, and when in contact with blood the collagen causes aggregation of platelets. This material is also resorbable within 14 to 56 days. Other products that can be used include bone wax, thrombin soaked gauze, and fibrin sealants.
Tranexamic acid 4.8% solution (Cyklokapron [Pharmacia & Upjohn Company]) is a topical antifibrinolytic that is commonly used to prevent excessive hemorrhage during surgery. The patient is instructed to rinse with 10 mL of the solution for 2 minutes, 4 times a day for 7 days. This adjunctive therapy is not necessary for routine treatment, but may be useful in cases involving multiple extractions.16 Preordering with the pharmacy is often necessary to be certain this drug is in stock.
ActCel (Coreva Health Sciences) and BloodSTOP (LifeScience PLUS) are new products made from specially treated sterilized cellulose that dissolves within 1 to 2 weeks of placement within the extraction socket. The material expands to 3 to 4 times its original size and is quickly converted to a gel. ActCel is hypoallergenic and bacteriostatic. The material is placed into the socket in a manner similar to Surgicel. ActCel works by a variety of mechanisms. It provides increasing surface area for clotting, enhances platelet aggregation, and makes calcium ions more available for the clotting cascade.25
CONCLUSION
 |
|
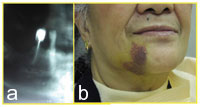
Figure 6. Preoperative radiograph of mandibular teeth requiring extraction. Even with meticulous surgical technique, a potential exists for minor complications following routine dentoalveolar surgery.
|
The recommendations for performing dental treatment on patients receiving anticoagulation medication have been clarified. Each patient must be evaluated individually, and the dentist should communicate often with the physician managing the anticoagulant therapy. Each case should be reviewed for the risk of bleeding during the dental procedure (Figure 6) if anticoagulation is continued versus the risk of thromboembolism if anticoagulation medication is reduced, altered, or stopped. Patients who require restorative procedures, periodontal scaling, or simple exodontia should continue their anticoagulant medication. When surgical procedures are more extensive, the dentist must be aware of the available hemostatic agents and keep in close contact with the patient to provide rapid follow-up if a complication occurs. It is advisable to check the pretreatment INR when bleeding is anticipated. Evidence indicates that for routine, in-office oral surgery procedures, anticoagulant medication should be continued if the INR is within the therapeutic range and proper local hemostatic measures are used.
References
1 Little JW, Falace DA, Miller CS, et al. Bleeding disorders. In: Dental Management of the Medically Compromised Patient. 6th ed. St Louis, Mo: Mosby; 2002.
2. Meehan S, Schmidt MC, Mitchell PF. The International Normalized Ratio as a measure of anticoagulation: significance for the management of the dental outpatient. Spec Care Dentist. 1997;17:94-96.
3. Troulis MJ, Head TW, Leclerc JR. Dental extractions in patients on an oral anticoagulant: a survey of practices in North America. J Oral Maxillofac Surg. 1998;56:914-917.
4. Abebe W. An overview of herbal supplement utilization with particular emphasis on possible interactions with dental drugs and oral manifestations. J Dent Hyg. 2003;77:37-46.
5. Ang-Lee MK, Moss J, Yuan CS. Herbal medi