Dental implants as tooth replacements have become a well-accepted mode of treatment with high rates of success and predictable outcomes. In recent years, much of the research by manufacturers and clinicians on dental implants has focused on fixture surfaces. The goal of new surface types, and/or surface microtextures, is to promote quicker and improved integration of the implant. While the possibilities of improving implantology along these lines are still being explored, a preliminary study suggests an almost twofold increase in bone-to-implant contact when using a microtextured implant surface.1 This goal is driven by patient demand for immediate tooth replacement. Accordingly, the direction of today’s progress in implant therapy is to expedite the entire process and thus meet our patients’ wishes.
Immediate implant placement has been well-documented as a sound clinical approach, with high success rates especially in the anterior region where implant form mimics the shape of the extraction socket.2 Threaded cylindrical and tapered root form implants work well and are often the clinician’s preference. A review of the literature reveals that very little has been written about immediate implants placed in the molar region after extraction. Fugazzotto3 concluded that immediate implant placement in molar extraction sites would succeed predictably with the aid of regenerative materials. Molar implants are usually placed in a delayed process where the extraction site has been allowed to heal for a period of 3 to 6 months, depending on the bone quality at the site.
Many authors advocate socket grafting to preserve ridge form.4 While this process appears to prevent collapse of the bony socket in the short term, long-term studies are not yet available to confirm the efficacy of this technique. Research by Lindhe, et al5 suggests that remodeling of the site will occur over the long-term. Lindhe’s research6 suggests that the implant should be placed more toward the lingual wall due to resorption effects on the buccal aspect.
Placing immediate implants in the molar region has 3 apparent obstacles. First, the size and shape of the socket differ markedly from the size and shape of an implant, and there is inherently a greater “jumping distance” the bone must cover from the wall of the extraction socket to reach the surface of the implant.7 Second, it can be very difficult to achieve primary or rigid fixation in a molar implant site due to resulting anatomy or morphology after the tooth has been extracted. Finally, because of occlusal forces and chewing habits in the posterior, this would put posterior implants at greater risk for macro- or micromovement, or premature “loading” due to mastication, and thus lead to fibrous integration and failure of osseointegration.
The purpose of this article is to propose a technique in which immediate molar implants can be placed routinely and predictably following extraction, ultimately reducing the time from extraction to final prosthesis. Indications for this treatment are molar teeth that can be extracted atraumatically, leaving bony cortical walls intact. Pre-existing infection is not a contraindication, unless it has caused severe alteration to the morphology of the site, such as the loss of a significant portion of buccal or lingual cortical bone.
METHOD
Proper informed consent must be obtained, including discussion of the options to graft the socket followed by traditional delayed placement. The patient is then appropriately anesthetized, and the tooth is removed atraumatically. Caution should be taken not to achieve so much vasoconstriction that there is insufficient bleeding in the socket, which would inhibit normal clot formation in the area.
 |
 |
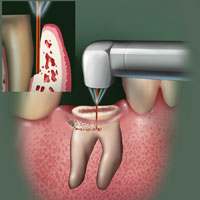
| Figure 1. Troughing around tooth with laser to aid in extraction. |
Figure 2. Use of the Er,Cr:YSGG to trough around and section roots. |
 |
 |
| Figure 3. Comparatively large defect that is produced when troughing with conventional carbide bur. | Figure 4. Root tip is luxated out. (Er,Cr:YSGG can remove ankylosed teeth with safety and minimal destruction of surrounding bone.) |
 |
 |
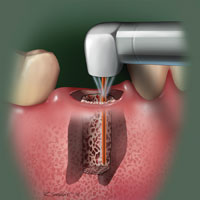
| Figure 5. Osteotomy is prepared with the Er,Cr:YSGG laser. | Figure 6. Implant is placed with primary stability achieved in the apical region. |
The Er,Cr:YSGG laser is used to aid in removing the tooth in most cases. It can be used to section the tooth and trough around the root tips (Figures 1 and 2). Use of the laser allows greater preservation of bone compared to conventional techniques such as the bur (Figure 3). The root can be traced with a 600-µm tip until the fragment can be accessed for luxation (Figure 4). In cases of ankylosis or complex root form, the laser can very accurately ablate the root structure directly, if it is unable to be luxated. Once the tooth has been removed, the socket is thoroughly de-epithelialized with the Er,Cr:YSGG. Any additional areas of abscess or pre-existing infection are enucleated and thoroughly debrided with the laser 0.5 mm past the visualized lesion boundary into the bone. After de-epithelialization, the cortical bone is perforated in a polka-dot pattern to induce “bleeders” throughout the socket in intervals approximately 1.0 mm apart.
The Er,Cr:YSGG laser is then used to initiate an osteotomy (Figure 5). This is where the Er,Cr:YSGG is of great benefit compared to conventional techniques. Traditionally, osteotomies are performed with burs, drills, or osteotomes. Molar extraction sockets present an anatomical form that makes it difficult to prepare an osteotomy. Traditional instruments such as burs and drills will slip, chatter, or misdirect from intended directional vectors. Additionally, vital anatomical structures, including the sinus and the mandibular canal, are in close proximity to molar sites and could be compromised. Using the laser, the osteotomy can be prepared precisely, conservatively, and without risk of harm to vital structures. The implant can then be placed with good primary stability.
In the author’s experience, the actual amount of bone-to-implant contact is not critical, and in most cases the implant appears to be “floating” in the socket from a lateral view on the periapical film. The only factor that matters is achieving good primary stability (Figure 6). A study by Scarano, et al8 indicates the importance of primary stability in achieving sufficient bone-to-implant contact. Another study by Lioubavina-Hack, Lang, and Karring9 indicates that primary instability will lead to fibrous integration, causing molar implants to fail. Ideal placement of the immediate molar implant is similar to principles used for placement of implants in healed-molar sites. It is based on prosthetically driven principles; it is also based on established biological principles of placement of immediate implants in the anterior region in terms of buccolingual and apicocoronal dimensions.
CASE REPORTS
Case 1
 |
 |
| Figure 7. Preoperative radiograph. Note bony defect on distal. |
Figure 8. Healing after 2 months. |
 |
 |
|
Figure 9. Four months postoperative. Final restoration in place. |
Figure 10. Final restoration on No. 19. |
The patient presented with a history of recent root canal therapy on tooth No. 19. There was a periodontal defect on the distal aspect with a 7.0-mm probing depth and a possible vertical crack. The tooth was symptomatic. The decay on the distal aspect of the tooth was at the level of the crest of the bone. The patient was given 3 options: crown lengthening followed by post and restoration with crown; extraction followed by delayed implant placement; or extraction followed by immediate im-plant placement. The patient opted for immediate implant placement. The tooth was surgically extracted without trauma to the bone due to use of the Er,Cr:YSGG laser. A Straumann 4.8 x 10 wide diameter platform implant was placed. Figures 7 to 10 show the periapical radiographs of the history and photos of the final result.
Case 2
 |
 |
| Figure 11. Preoperative radiograph. |
Figure 12. One year postoperative, upon the patient’s return from Africa. |
The patient presented with failing endodontic treatment on tooth No. 19. The patient is a diplomat and primarily resides in Africa, and is only in the United States for 2 weeks during December each year. Teeth Nos. 19, 30, and 31 all required extraction and immediate implants due to failing endo. A porcelain-metal, winged, Maryland bridge-type prosthesis was made to temporarily replace tooth No. 19 and allow the pa-tient to chew while the implant was osseointegrating. Teeth Nos. 30 and 31 were not loaded, and no temporary prosthesis was made for the lower right side. The patient returned in approximately 1 year. The Maryland bridge was removed, a solid abutment was torqued to 30 Ncm, and a definitive implant crown was fabricated (Figures 11 to 12).
Case 3
 |
 |
| Figure 13. Preoperative radiograph. | Figure 14. Five months postoperative. |
The patient presented with failing endodontic treatment on tooth No. 30. The tooth was extracted following the laser-assisted protocol as described in Method, and a permanent prosthesis was placed 6 months after the extraction. Further remodeling of the socket is expected over the next several months. (Figures 13 to 14).
Case 4
 |
 |
| Figure 15. Preoperative radiograph, tooth No. 19. | Figure 16. Intraoperative radiograph. |
 |
 |
| Figure 17. Seven months postoperative; treatment was delayed while patient recovered from carcinoma of the thyroid. | Figure 18. Seven-month postoperative photograph. |
The patient presented with failed endodontic treatment on teeth Nos. 13, 19, and 30. Tooth No. 30 was retreated with conventional endodontic therapy. Immediate implants were placed into the extraction sockets of teeth Nos. 13 and 19. Tooth number 13 was immediately loaded with a provisional restoration. Both implants integrated successfully; tooth No. 13 received a definitive restoration at 3 months and tooth No. 19 at 7 months. The patient discovered she had carcinoma of her thyroid gland 4 months after implant placement. Implant therapy continued simultaneously with cancer treatment. Figures 15 to 18 show the tissue health around the molar implant at 3 months. Permanent abutments and crowns were also placed at this time.
DISCUSSION
This protocol for implant placement uses principles from well-proven techniques and biological studies. Relevant principles include those of the healing of extraction sites. In a study by Cardaropoli, Araújo, and Lindhe,10 healing was found to occur in the following stages: (1) blood clots fill the extraction site, (2) provisional connective tissue matrix replaces the blood clot, (3) woven bone fills in most of the provisional matrix, (4) cortical bone grows over the woven bone, and (5) bone marrow fills the inside of the lamellar bone.
The consensus in the literature11 is that the bone “jumping distance” in an extraction site will predictably reach 1.5 mm, with 2 mm being the maximum before results become unpredictable and thus useless to the clinician. Immediate molar implants necessitate jumping distances of greater than 2 mm. The author has observed the bone to jump a distance of up to 3 mm on a predictable basis. While more research is necessary to corroborate this finding, evidence in the literature supports the possibility of such a wide jump distance. Furthermore, Fugazzotto3 indicates that immediate molar implants are possible and succeed predictably even with conventional techniques; we assert that the laser makes the procedure easier for the practitioner and contributes in other ways that make immediate placement in molar sites a viable treatment.
The feasibility of this technique can at one level be drawn from what is known about the lateral ridge expansion technique (LRE), which is well-documented.12 When a LRE technique is used, the bone jump distance in a mesiodistal direction is often more than 2 mm.
On another level, the success of immediate molar implants can be partially attributed to the use of the Er,Cr:YSGG laser. A study in rats by Kesler, Romanos, and Koren13 using an Er:YAG laser showed better osseointegration and faster bone healing than that achieved after conventional techniques using a rotary drill. The YSGG and YAG lasers have similar wavelengths: 2,790 nm and 2,940 nm respectively, so it is reasonable to presume that the two will have similar effects, although more clinical data are necessary to prove this. A possible cause for this is the bactericidal effect of laser irradiation on extraction sites. A number of published studies indicate that laser irradiation lowers the bacteria count in the affected area.14 Periodontal pathogens in the site can restrict osteogenesis,15 so lasers may aid in bone growth by reducing the bacteria that hamper it. Furthermore, laser surgery causes less damage to the surrounding areas.16 Regeneration of bone in the osteotomy site may then occur faster, with fewer original, compromised collagen fibers to regrow before osteogenesis can begin.
The other factor contributing to the success of this procedure is the stage of bone healing that takes place when the implant is installed and when loading begins. Conventional techniques dictate that a molar implant be placed in the socket at least 3 months after extraction to allow it to heal. However, in the first 4 months after extraction, substantial buccolingual and apicocoronal ridge reduction occurs, and osteogenesis slows down after 2 months.17 Placing an implant immediately does not allow bone resorption to occur and prevents soft connective tissue from forming instead of hard bone.18 The advantages of immediate placement and the complications it alleviates in the anterior region are well-documented; the case is similar for the posterior region, although the success of immediate implant placement is attributable to other factors as well.
CONCLUSION
Acknowledgment
The author wishes to thank Daniel Shapero for assistance in researching this article.
References
- Todisco M, Trisi P. Histomorphometric evaluation of six dental implant surfaces after early loading in augmented human sinuses. J Oral Implantol. 2006;32:153-166.
- Chen ST, Wilson TG Jr, Hämmerle CH. Immediate or early placement of im-plants following tooth extraction: review of biologic basis, clinical procedures, and outcomes. Int J Oral Maxillofac Implants. 2004;19(suppl):12-25.
- Fugazzotto PA. Implant placement at the time of maxillary molar extraction: technique and report of preliminary results of 83 sites. J Periodontol. 2006;77:302-309.
- Sclar AG. Preserving alveolar ridge anatomy following tooth removal in conjunction with immediate implant placement. The Bio-Col technique. Atlas Oral Maxillofac Surg Clin North Am. 1999;7:39-59.
- Araujo MG, Sukekava F, Wennstrom JL, et al. Ridge alterations following implant placement in fresh extraction sockets: an experimental study in the dog. J Clin Periodontol. 2005;32:645-652.
- Araujo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32:212-218.
- Botticelli D, Berglundh T, Buser D, et al. The jumping distance revisited: an experimental study in the dog. Clin Oral Implants Res. 2003;14:35-42.
- Scarano A, Degidi M, Iezzi G, et al. Correlation between implant stability quotient and bone-implant contact: a retrospective histological and histomorphometrical study of seven titanium implants retrieved from humans. Clin Implant Dent Relat Res. 2006;8:218-222.
- Lioubavina-Hack N, Lang NP, Karring T. Significance of primary stability for osseointegration of dental implants. Clin Oral Implants Res. 2006;17:244-250.
- Cardaropoli G, Araújo M, Lindhe J. Dynamics of bone tissue formation in tooth extraction sites: an experimental study in dogs. J Clin Periodontol. 2003;30:809-818.
- Wilson TG Jr, Schenk R, Buser D, et al. Implants placed in immediate extraction sites: a report of histologic and histometric analyses of human biopsies. Int J Oral Maxillofac Implants. 1998;13:333-341.
- Scipioni A, Bruschi GB, Calesini G, et al. Bone regeneration in the edentulous ridge expansion technique: histologic and ultrastructural study of 20 clinical cases. Int J Periodontics Restorative Dent. 1999;19:269-277.
- Kesler G, Romanos G, Koren R. Use of Er:YAG laser to improve osseointegration of titanium alloy implants – a comparison of bone healing. Int J Oral Maxillofac Implants. 2006;21:375-379.
- Türkün M, Türkün LS, Çelik EU, et al. Bactericidal effect of Er,Cr:YSGG laser on Streptococcus mutans. Dent Mater J. 2006;25:81-86.
- Loomer PM, Sigusch B, Sukhu B, et al. Direct effects of metabolic products and sonicated extracts of Porphyromonas gingivalis 2561 on osteogenesis in vitro. Infect Immun. 1994;62:1289-1297.
- Walsh JT Jr, Flotte TJ, Deutsch TF. Er:YAG laser ablation of tissue: effect of pulse duration and tissue type on thermal damage. Lasers Surg Med. 1989;9:314-326.
- Evian CI, Rosenberg ES, Coslet JG, et al. The osteogenic activity of bone removed from healing extraction sockets in humans. J Periodontol. 1982;53:81-85.
- Ferrara A, Galli C, Mauro G, et al. Immediate provisional restoration of postextraction implants for maxillary single-tooth replacement. Int J Periodontics Restorative Dent. 2006;26:371-377.
Dr. Singer has a private practice in the Washington, DC, and Alexandria, Va, areas. He is an assistant clinical professor in the Department of Surgery at George Washington University. He can be reached by e-mailing novasmiles@yahoo.com.











