INTRODUCTION
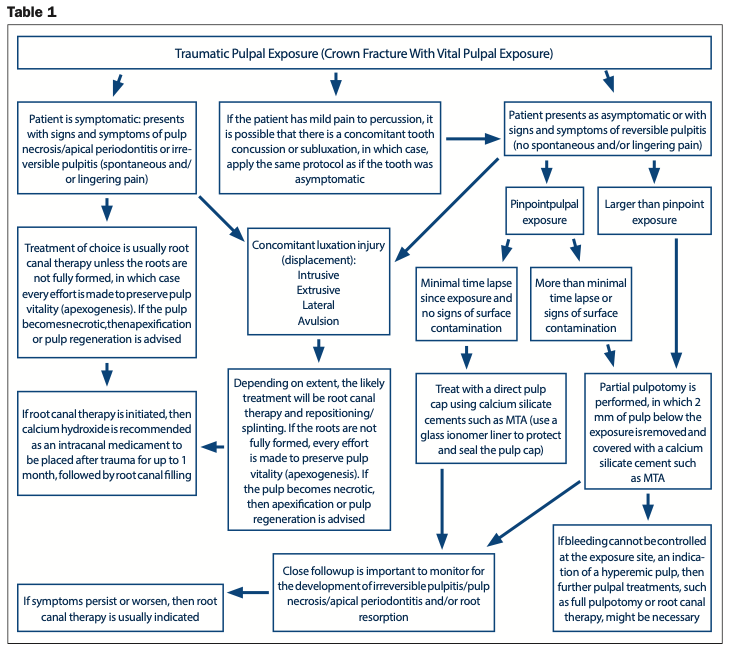
There are 5 important questions to consider prior to treating a traumatic pulpal exposure (see Table 1):
Question 1: How long has the exposure been present?
Short answer: The longer the exposure has been present, the higher the likelihood for bacterial contamination of the pulp. However, as long as the pulp is still vital and the patient’s symptoms don’t necessitate root canal therapy, vital pulpal treatments can be utilized.
Question 2: Is the patient symptomatic? What are the pulpal and periapical diagnoses?
Short answer: The pulpal and periapical diagnoses will dictate treatment. For example, if the pulpal diagnosis is necrosis or irreversible pulpitis (spontaneous or lingering pain), then pulpectomy (root canal therapy) is usually indicated rather than a vital pulpal treatment, such as a pulp cap or partial pulpotomy. A tooth with periapical pathology is obviously not a good candidate for vital pulpal treatments. A proper diagnosis must be reached before deciding to perform vital pulpal treatments. Vital pulpal treatments will usually not work if the pulp is severely inflamed or infected.
Question 3: Is there a concomitant luxation injury or root fracture?
Short answer: A concomitant luxation injury (see Tables 1 to 3) or root fracture might further complicate pulpal circulation and thereby necessitate a full root canal rather than vital pulpal treatment.
Question 4: Are the roots fully developed?
Short answer: As a general rule, we make every effort to preserve the pulp vitality of an immature tooth to allow for continued root growth (apexogenesis). It is important to remember that although vital pulpal treatments are usually the treatment of choice for immature permanent teeth, they can be performed for both mature and immature permanent teeth.
Question 5: How large is the fracture/exposure?
Short answer: There are different levels of vital pulpal therapy, ranging from a direct pulp cap to a full pulpotomy. The size of the exposure will play a role in dictating which vital pulpal treatment is best. There are times when a fracture might compromise significant tooth structure, necessitating a post (for retention of a core). In this case, endodontic treatment is indicated even when the pulp is vital and the exposure is relatively small.
DEFINITION
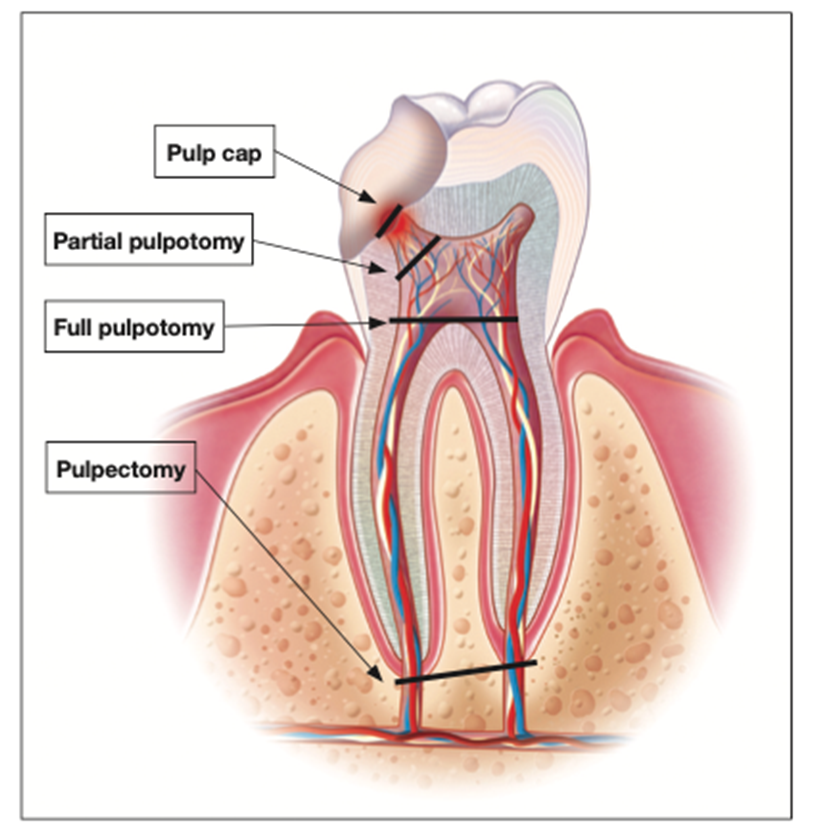
Vital pulp therapy (VPT) techniques are means of preserving the vitality and function of the dental pulp after injury resulting from trauma, caries, or restorative procedures. VPT procedures have traditionally included indirect or direct pulp capping and partial or complete pulpotomy1 (Figure 1). VPT includes the removal of part of the pulp, allowing the rest of it to remain vital and functional. We assume that by removing a small portion of the superficial inflamed pulp, the remaining pulp remains healthy.
When the pulp is exposed, bacterial invasion can be resisted by both the patient’s immune system present within the pulp and the bathing of the area by the patient’s saliva, which prevents impaction of contaminated debris/bacteria. While the pulp is a low-compliant environment, making it more vulnerable to injury, it also has the ability to heal itself. Vital pulpal therapy is built upon this very important principle. Overall, VPT has been shown to have a very high success rate.2
OBJECTIVES
There are 2 essential objectives for the treatment of the vital pulp that make the level of pulpal amputation important: A wound dressing should be placed on non-inflamed tissue, and the loss of tooth structure should be kept to a minimum. In crown-fractured teeth showing vital pulp tissue after exposure, not more than 2 mm of the pulp beneath the exposure needs to be removed3 (Figures 1 and 2).
CVEK PULPOTOMY
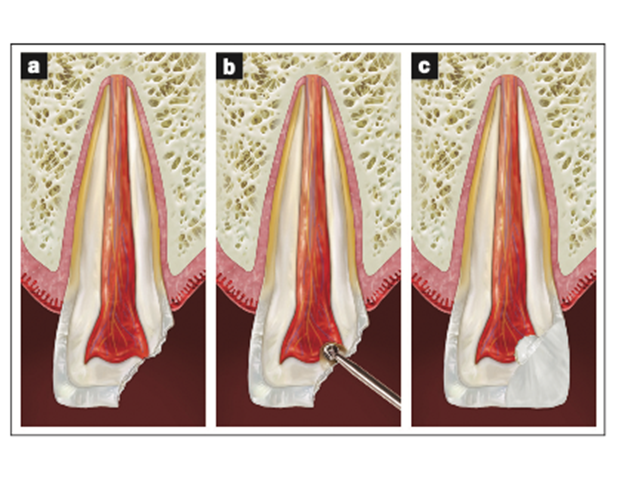
With children and young adults, most teeth that have the pulp exposed by a crown fracture can be treated successfully by partial pulpotomy, also known as a Cvek pulpotomy, in which a small portion of the pulp tissue (usually 2 mm) is removed beneath an exposure4 (Figure 2 and Table 2). The major benefit to maintaining vital pulp in immature teeth is that it allows for continued root growth (apexogenesis). The exposure size amenable to partial pulpotomy will usually range from 0.5 to 4 mm in size.5
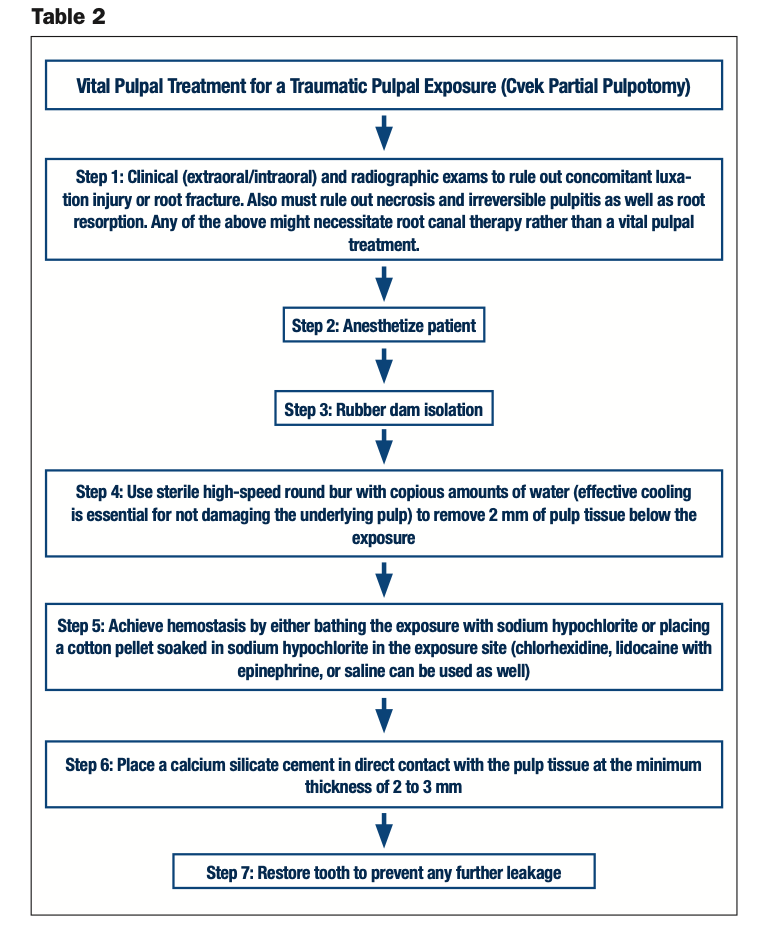
This treatment can also be performed on mature permanent teeth, not just children’s.4,6 Some studies show a higher success rate with VPTs on an immature permanent tooth when compared to a mature permanent tooth with a closed apex.7 The reason is that, in older patients, the pulp is more fibrotic, is less cellular with a reduced blood supply, and has a diminished healing capacity.8,9 It is also impractical to do vital pulpal treatment on a fully mature tooth that you anticipate will end up needing full root canal treatment, as future entry into the root canal will be made more difficult once vital pulpal therapy is done. Cvek,5 in a clinical report of partial pulpotomies carried out on 60 young teeth, found a success rate of 96.7%, with the majority of these teeth being treated within 4 days of traumatic exposure. In another classic study by Cvek,4 partial pulpotomies were performed on 178 teeth with traumatic pulpal exposures. Patients were followed for 3 to 15 years, both clinically and radiographically. Healing with hard-tissue barriers and the maintaining of pulp vitality were seen in 95% of the cases. A majority of these cases were treated within 3 days of the trauma.
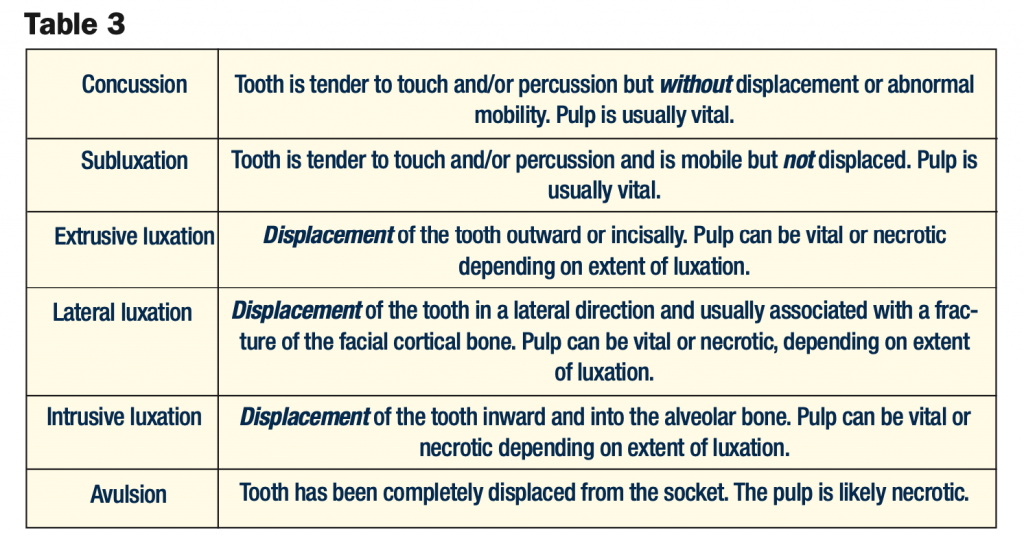
PARTIAL PULPOTOMY VS DIRECT PULP CAP
The advantage of partial pulpotomy (removing 2 mm of coronal pulp) compared to pulp capping lies in better control of the surgical wound and retention of the sealing material. Capping of the pulp is recommended only when the exposure is small (less than 1 mm in size) and when it can be treated shortly after the accident.10 These indications apply to only a limited number of teeth, and in the majority of cases, a partial pulpotomy is therefore performed.5 It is also noteworthy that most studies find a much higher success rate with partial pulpotomy compared to pulp capping after a traumatic pulpal exposure11-13 due to the better control of superficial inflammation below the exposure. It has been clearly shown that as time elapses (from one hour to 7 days) after injury, the success of pulp capping significantly decreases (from 93% to 56%).14 Vital pulpal treatments can be used as a permanent treatment modality for mature and immature permanent teeth with complicated crown fractures, and re-entering of the pulp at a later time is not necessary as long as the pulp remains vital.7,15
HEMOSTASIS
Sodium hypochlorite is an antimicrobial solution that provides hemostasis, disinfection of the dentin pulp interface, biofilm removal, chemical removal of the blood clot and fibrin, and clearance of dentinal chips along with damaged cells at the mechanical exposure site. Therefore, sodium hypochlorite can be used to disinfect the exposure site during a pulpotomy prior to restoring the pulpal wound (saline or chlorhexidine can be used as well).16 A cotton-soaked pellet is placed on the pulpal wound with pressure for this purpose.
The examination of pulp tissues after exposure is a critical step in pulp assessment. Controlled bleeding (complete hemostasis) is needed for a successful partial pulpotomy. If hemostasis cannot be achieved, this is an indicator that the pulp is inflamed and should not be “capped”. On occasion, more pulp tissue needs to be removed to achieve hemostasis, and what was initially planned as a partial pulpotomy might turn into a full pulpotomy. A pulpal diagnosis of necrosis (non-vital pulp) or the inability to achieve hemostasis would obviously preclude vital pulpal treatments such as a pulp cap or pulpotomy (partial or full) (Figure 3).17
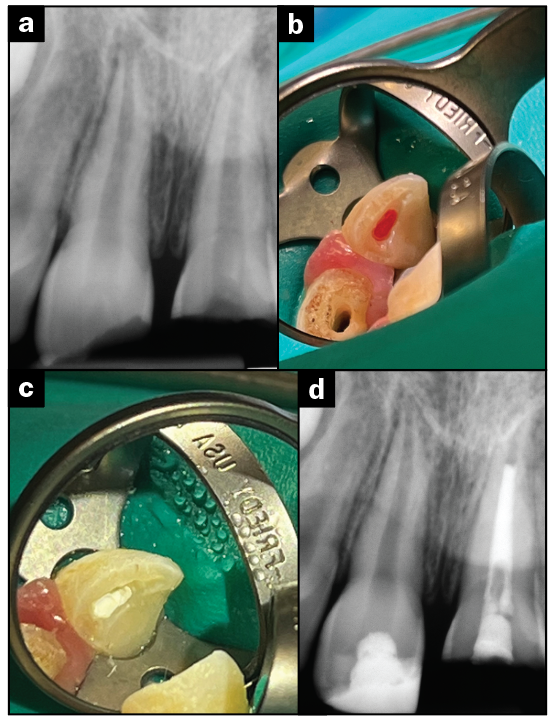
CALCIUM SILICATE CEMENTS
There have been few materials as groundbreaking in endodontics as mineral trioxide aggregate (MTA). This material, developed in 1993, was the first of the calcium silicate cements (CSCs) and was initially introduced for the purpose of sealing root perforations. Over time, they were found to be so biocompatible and successful in their applications that they have become mainstreamed for the use of apexogenesis, apexification, apicoectomies, and to seal root perforations (see Table 4).18-20
CSCs are a class of materials that include tricalcium silicates, dicalcium silicates, hydraulic calcium silicate cements, and “bioceramics” (MTA [Angelus], TheraCal [BISCO Dental Products], EndoSequence BC RRM [Brasseler USA], EndoSequence BC sealer [Brasseler USA], and Biodentine [Septodont] are some examples). CSCs have become more commonplace for use in VPT procedures. These materials are placed directly on the pulpal wound (Figure 4). While calcium hydroxide (CH) has classically been used for vital pulpal treatments, most current studies clearly demonstrate that CSCs are superior in terms of healing and success rates and are the preferred materials to place on a pulpal exposure (once hemostasis is achieved).4,21-24 MTA was the first of these materials introduced for this purpose, but newer materials have recently flooded the market. One of the advantages of the newer materials is that they don’t cause significant tooth discoloration, which can be beneficial, especially when used in the aesthetic zone.
The mechanism by which CH works is by eliciting the formation of a dentinal bridge right below the capping material. These materials cause superficial coagulation necrosis (1 to 1.5 mm in depth), which induces a low-grade irritation that leads to differentiation of the undifferentiated cells of the pulp. These cells then synthesize predentin, which is subsequently mineralized, while the coagulated tissue is calcified. Finally, predentin is transformed into dentin by further mineralization. In essence, it is the low-grade irritation from coagulation necrosis that causes the hard-tissue barrier to form right below the capping material. This low-grade irritation is not strong enough to damage the pulp. Still, it is sufficient to elicit an immune response that triggers the formation of a dentin bridge, which protects the underlying pulp. CSCs such as MTA benefit from forming a stronger dentin bridge without the disadvantage of triggering a zone of necrosis, like in the case with CH. Pitt Ford et al23 compared CH and MTA and found that the majority of pulps that were capped with MTA were free of inflammation, and all of them showed calcified bridge formations after 5 months. In contrast, the pulp of teeth that were capped with CH showed the presence of inflammation and significantly less calcified bridge formation. Tziafas et al25 found that during the first 2 weeks, osteodentin matrix formation takes place right below the MTA, and after 3 weeks, a complete layer of reparative dentin (tertiary dentin) is formed at the capping site. Therefore, the placement of MTA on pulp tissue causes proliferation, migration, and differentiation of odontoblast-like cells that produce a collagen matrix, which then mineralizes and produces osteodentin. This is then replaced with a tertiary dentinal bridge a few weeks after pulp capping.18,23,25-33
CSCs offer many advantages as agents for use in pulpotomies. They are biocompatible, provide excellent resistance to microleakage, allow an opportunity for dentin bridging at the site of pulp exposure, are dimensionally stable over time, and appear to be associated with very positive clinical outcomes. The main difference between CSCs and CH may be that CSCs provide a good protective barrier against microleakage and do not break down, requiring replacement, as is the case with CH. CSCs form a much higher quality dentin bridge than CH. If microleakage occurs in the case of CH, bacteria can reach the dentin bridge, which usually has numerous tunneling defects, allowing for potential bacterial leakage directly into the pulp.3,34 This is not significant when CSCs are used as the dentin bridge formed does not have tunneling defects.23,35
CONCLUSION
When treatment planned correctly, vital pulpal treatments, such as a partial pulpotomy, can be a valuable option with a high success rate. They can be successfully utilized not only for traumatic pulpal exposures but for carious pulpal exposures as well.36-38 The key to this high success rate is proper diagnosis and a well-sealed restoration above the capping material. Bacterial leakage ultimately can cause any vital pulpal treatment to fail.5,26,27,39,40
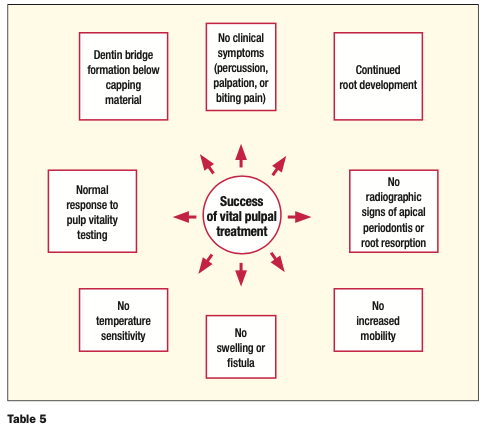
A successful followup to VPT would be dentin bridge formation, continued root development, positive response to pulp vitality testing, no symptoms, and no radiographic development of apical periodontitis or root resorption (see Table 5).
ACNOWLEDGEMENTS
The author wishes to thank Drs. Charles Solomon, Leslie Elfenbein, and Eric Wachs for their valuable input and guidance.
REFERENCES
1. AAE Position Statement on Vital Pulp Therapy. J Endod. 2021;47(9):1340–44. doi:10.1016/j.joen.2021.07.015
2. Sabeti M, Huang Y, Chung YJ, et al. Prognosis of vital pulp therapy on permanent dentition: a systematic review and meta-analysis of randomized controlled trials. J Endod. 2021;47(11):1683–95. doi:10.1016/j.joen.2021.08.008
3. Cvek M, Cleaton-Jones PE, Austin JC, et al. Pulp reactions to exposure after experimental crown fractures or grinding in adult monkeys. J Endod. 1982;8(9):391–7. doi:10.1016/S0099-2399(82)80092-7
4. Cvek M. Partial pulpotomy in crown fractured incisors: results 3 to 15 years after treatment. Acta Stomatol Croat. 1993;27:167–73.
5. Cvek M. A clinical report on partial pulpotomy and capping with calcium hydroxide in permanent incisors with complicated crown fracture. J Endod. 1978;4(8):232–7. doi:10.1016/S0099-2399(78)80153-8
6. Lin LM, Ricucci D, Saoud TM, et al. Vital pulp therapy of mature permanent teeth with irreversible pulpitis from the perspective of pulp biology. Aust Endod J. 2020;46(1):154-166. doi:10.1111/aej.12392
7. Bimstein E, Rotstein I. Cvek pulpotomy—revisited. Dent Traumatol. 2016;32(6):438-442. doi:10.1111/edt.12297
8. Fong CD, Davis MJ. Partial pulpotomy for immature permanent teeth, its present and future. Pediatr Dent. 2002;24(1):29-32.
9. Massler M. Therapy conductive to healing of the human pulp. Oral Surg Oral Med Oral Pathol. 1972;34(1):122–30. doi:10.1016/0030-4220(72)90281-2
10. Heide S, Kerekes K. Delayed direct pulp capping in permanent incisors of monkeys. Int Endod J. 1987;20(2):65-74. doi:10.1111/j.1365-2591.1987.tb00591.x
11. Fuks AB, Bielak S, Chosak A. Clinical and radiographic assessment of direct pulp capping and pulpotomy in young permanent teeth. Pediatr Dent. 1982;4(3):240–4.
12. Barthel CR, Rosenkranz B, Leuenberg A, et al. Pulp capping of carious exposures: treatment outcome after 5 and 10 years: a retrospective study. J Endod. 2000;26(9):525–8. doi:10.1097/00004770-200009000-00010
13. Dammaschke T, Leidinger J, Schäfer E. Long-term evaluation of direct pulp capping–treatment outcomes over an average period of 6.1 years. Clin Oral Investig. 2010;14(5):559–67. doi:10.1007/s00784-009-0326-9
14. Cox CF, Bergenholtz G, Fitzgerald M, et al. Capping of the dental pulp mechanically exposed to the oral microflora—a 5-week observation of wound healing in the monkey. J Oral Pathol. 1982;11(4):327–39. doi:10.1111/j.1600-0714.1982.tb00173.x
15. Cvek M, Lundberg M. Histological appearance of pulps after exposure by a crown fracture, partial pulpotomy, and clinical diagnosis of healing. J Endod. 1983;9(1):8-11. doi:10.1016/S0099-2399(83)80005-3
16. Hafez AA, Cox CF, Tarim B, et al. An in vivo evaluation of hemorrhage control using sodium hypochlorite and direct capping with a one- or two-component adhesive system in exposed nonhuman primate pulps. Quintessence Int. 2002;33(4):26–72.
17. Cho SY, Seo DG, Lee SJ, et al. Prognostic factors for clinical outcomes according to time after direct pulp capping. J Endod. 2013;39(3):327–31. doi:10.1016/j.joen.2012.11.034
18. Parirokh M, Torabinejad M. Mineral trioxide aggregate: a comprehensive literature review—Part III: Clinical applications, drawbacks, and mechanism of action. J Endod. 2010;36(3):400–13. doi:10.1016/j.joen.2009.09.009
19. Parirokh M, Torabinejad M. Mineral trioxide aggregate: a comprehensive literature review—Part I: chemical, physical, and antibacterial properties. J Endod. 2010;36(1):16-27. doi:10.1016/j.joen.2009.09.006
20. Torabinejad M, Parirokh M. Mineral trioxide aggregate: a comprehensive literature review—part II: leakage and biocompatibility investigations. J Endod. 2010;36(2):190-202. doi:10.1016/j.joen.2009.09.010
21. Bakland LK, Andreasen JO. Will mineral trioxide aggregate replace calcium hydroxide in treating pulpal and periodontal healing complications subsequent to dental trauma? A review. Dent Traumatol. 2012;28(1):25-32. doi:10.1111/j.1600-9657.2011.01049.x
22. Mente J, Geletneky B, Ohle M, et al. Mineral trioxide aggregate or calcium hydroxide direct pulp capping: an analysis of the clinical treatment outcome. J Endod. 2010;36(5):806–13. doi:10.1016/j.joen.2010.02.024
23. Pitt Ford TR, Torabinejad M, Abedi HR, et al. Using mineral trioxide aggregate as a pulp-capping material. J Am Dent Assoc. 1996;127(10):1491–4. doi:10.14219/jada.archive.1996.0058
24. Faraco IM Jr, Holland R. Response of the pulp of dogs to capping with mineral trioxide aggregate or a calcium hydroxide cement. Dent Traumatol. 2001;17(4):163–6. doi:10.1034/j.1600-9657.2001.170405.x
25. Tziafas D, Pantelidou O, Alvanou A, et al. The dentinogenic effect of mineral trioxide aggregate (MTA) in short-term capping experiments. Int Endod J. 2002;35(3):245–54. doi:10.1046/j.1365-2591.2002.00471.x
26. Fuks AB, Cosack A, Klein H, et al. Partial pulpotomy as a treatment alternative for exposed pulps in crown-fractured permanent incisors. Endod Dent Traumatol. 1987;3(3):100–2. doi:10.1111/j.1600-9657.1987.tb00610.x
27. de Blanco LP. Treatment of crown fractures with pulp exposure. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1996;82(5):564–8. doi:10.1016/s1079-2104(96)80204-6
28. Foreman PC, Barnes IE. Review of calcium hydroxide. Int Endod J. 1990;23(6):283–97. doi:10.1111/j.1365-2591.1990.tb00108.x
29. Olsburgh S, Jacoby T, Krejci I. Crown fractures in the permanent dentition: pulpal and restorative considerations. Dent Traumatol. 2002;18(3):103–15. doi:10.1034/j.1600-9657.2002.00004.x
30. Schröder U, Granath LE. Early reaction of intact human teeth to calcium hydroxide following experimental pulpotomy and its significance to the development of hard tissue barrier. Odontol Revy. 1971;22(4):379–95.
31. Schröder U. Reaction of human dental pulp to experimental pulpotomy and capping with calcium hydroxide. Odontol Revy. 1973;24:(Suppl 25):1–97.
32. Cvek M, Granath L, Cleaton-Jones P, et al. Hard tissue barrier formation in pulpotomized monkey teeth capped with cyanoacrylate or calcium hydroxide for 10 and 60 minutes. J Dent Res. 1987;66(6):1166–74. doi:10.1177/00220345870660061501
33. Kuratate M, Yoshiba K, Shigetani Y, et al. Immunohistochemical analysis of nestin, osteopontin, and proliferating cells in the reparative process of exposed dental pulp capped with mineral trioxide aggregate. J Endod. 2008;34(8):970–4. doi:10.1016/j.joen.2008.03.021
34. Cox CF, Sübay RK, Ostro E, et al. Tunnel defects in dentin bridges: their formation following direct pulp capping. Oper Dent. 1996;21(1):4-11.
35. Nair PN, Duncan HF, Pitt Ford TR, et al. Histological, ultrastructural and quantitative investigations on the response of healthy human pulps to experimental capping with mineral trioxide aggregate: a randomized controlled trial. Int Endod J. 2008;41(2):128–50. doi:10.1111/j.1365-2591.2007.01329.x
36. Elmsmari F, Ruiz XF, Miró Q, et al. Outcome of partial pulpotomy in cariously exposed posterior permanent teeth: a systematic review and meta-analysis. J Endod. 2019;45(11):1296-1306.e3. doi:10.1016/j.joen.2019.07.005
37. Li Z, Cao L, Fan M, et al. Direct pulp capping with calcium hydroxide or mineral trioxide aggregate: a meta-analysis. J Endod. 2015;41(9):1412–7. doi:10.1016/j.joen.2015.04.012
38. Mejàre I, Cvek M. Partial pulpotomy in young permanent teeth with deep carious lesions. Endod Dent Traumatol. 1993;9(6):238–42. doi:10.1111/j.1600-9657.1993.tb00279.x
39. Fuks AB, Gavra S, Chosack A. Long-term follow-up of traumatized incisors treated by partial pulpotomy. Pediatr Dent. 1993;15(5):334–6.
40. Parirokh M, Torabinejad M, Dummer PMH. Mineral trioxide aggregate and other bioactive endodontic cements: an updated overview—part I: vital pulp therapy. Int Endod J. 2018;51(2):177-205. doi:10.1111/iej.12841
ABOUT THE AUTHOR
Dr. Stern is a Diplomate of the American Board of Endodontics. He is the director of endodontics at the Touro College of Dental Medicine and frequently lectures on clinical endodontics. He has lectured at local county dental societies, the New Jersey Dental Association Annual Session, and the Greater New York Dental Meeting. He maintains a private practice, Clifton Endodontics, in Clifton, NJ. He can be reached at [email protected] or via the Instagram handle @the_barbed_broach1.
Disclosure: Dr. Stern reports no disclosures.


