Adhesive resins are designed to provide strong coupling between resin composites and enamel and dentin. The earliest dental adhesives were relatively hydrophobic and were placed directly on enamel and dentin smear layers1 (Figure 1a), although the presence of these layers was unknown at that time. Today, that technique could be described as the “no-etch” bonding technique. The low bond strength produced by these early adhesives was because that the adhesives could not penetrate the smear layer (Figure 1b), and they could not form resin tags. When the bonds were stressed to failure, the fractures occurred within the smear layer (Figure 1b). That is, the apparent bond strength was actually the intrinsic, cohesive strength of the smear layer (approximately 5 to 10 MPa) rather than the strength of the resin-dentin bonds. These low strengths were less than the polymerization stresses (approximately 15 to 17 MPa) that develop during light-curing resin composites.2 This often led to debonding of 1 or more sides of a restored cavity, causing postoperative sensitivity, marginal staining, and secondary caries.
 |
| Figures 1a and 1b. a. Scanning electron micrograph of the fractured edge of smear layer-covered human dentin. Note the presence of smear plugs occluding the orifices of the tubules. b. Adhesive resin (blue) bonded to smear layer (no-etch bonding systems). When stressed to failure, the split occurs at the smear layer (black gap), indicating that it was the weak link when using the no-etch technique. |
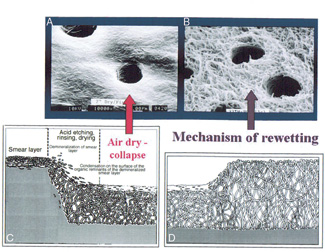 |
| Figures 2a through 2d. Schematic showing that smear layers are made up of microscopic cutting debris burnished on dentin. Total etching not only removes the smear layer, it also removes all of the mineral from around collagen fibrils in the matrix. After rinsing, the spaghetti-like collagen fibrils are suspended in water. If the surface is dried, the fibrils collapse upon themselves, decreasing the size of interfibrillar spaces that are necessary for resin uptake (modified from Pashley et al.5). The water used in wet bonding or in primers re-expands the collapsed matrix when the total-etch technique is coupled with wet bonding, preparing it for resin uptake. Courtesy of Dr. Franklin Tay, University of Hong Kong, with permission. |
If stronger resin-tooth bonds were to be achieved, the smear layers would have to be removed. Fortunately, smear layers are very acid-labile. Buonocore3 found that acid-etching enamel caused a large increase in resin-enamel bond strength (approximately 20 to 25 MPa). At that time, the presence of the smear layer was not known. However, Buonocore knew that acid etching removed an unidentified factor that interfered with strong resin-enamel bonds. The success of bonding resins to acid-etched enamel is because of the fact that enamel contains little protein, and it can be dried without causing any collapse of the roughened surface. When Buonocore et al4 tried those same procedures on dentin, they were disappointed to find that the resin-dentin bond strengths to acid-etched dentin were very low (5 to 10 MPa) and were about the same as resin-dentin bonds made to smear layer-covered dentin. They did not know that acid-etching dentin transformed the surface from a hard, mineralized surface to a very soft, mineral-free, collagen-rich surface that collapses when air-dried (Figures 2a through 2d). Although it is now known that acid-etching removes the smear layer and smear plugs, thereby making the tubules available for resin tag formation, drying the surface collapses the spaghetti-like collagen fibrils, making resin infiltration difficult.5
Over the next 20 years, little progress was made in dentin bonding. In 1982, Nakabayashi et al6 published their classic paper on how resin infiltration of acid-etched dentin completely transforms the surface from one that is crystalline, acid-sensitive, and relatively hydrophilic to a structure that is organic, acid-resistant, and relatively hydrophobic. They showed that following acid etching and water rinsing, the mineral phase of dentin was removed. All that was left of the original dentin was the collagenous matrix. The solubilized mineral phase was extracted, but was replaced by resin during resin infiltration. The new surface was neither dentin nor resin, but a hybrid of both. They coined the term “hybrid layer” to identify the newly formed bioengineered structure. The hybrid layer is very strong and tough7 when properly formed and affords significant micromechanical retention for resin composites. The paper by Nakabayashi and colleagues6 provided new insight and a rationale that explained how resin-tooth bonding occurred.
 |
| Figures 3a through 3c. In the 3-bottle total-etch bonding technique, acid is used to remove the smear layer and smear plugs (seen in Figure 1) from the dentin and to remove peritubular dentin, increasing the diameter of the tubules (a) that will result in stronger resin tags. The etched surface is then primed with dilute methacrylates (purple) in a volatile solvent (b) to ensure that the matrix is expanded and precoated with methacrylates. Then a solvent-free adhesive (green) is applied that diffuses into the primed surface and down into the tubules, where it polymerizes with the primer (c). Modified from Bouillaguet10 with permission. |
Two years earlier, Fusayama8 advocated etching both enamel and dentin simultaneous (ie, the total-etch concept). This was a very controversial concept in the United States and Europe, where acid-etching dentin was thought to produce pulpal death. However, a careful review of the literature indicated that the pulpal reactions that occurred following acid etching were largely because of inadequate sealing of etched cavities, and were the consequence of bacterial leakage.9 Over the past decade, reduction in etching time and improvements in bonding formulations and techniques have eliminated the acid-etching controversy. Because the total-etch technique led to collapse of the demineralized matrix when it was air-dried, the use of primer pretreatments was necessary to re-expand the matrix prior to applying the final bonding agent. Bonding became a 3-step process: (1) total etch and rinse (Figure 3a), (2) priming and evaporation of solvent (Figure 3b), and (3) application of the adhesive followed by light curing (Figure 3c).10
Another major advance in dentin bonding technique that occurred in the early 1990s was the “wet” (or moist) bonding technique developed by Kanca.11-13 Prior to the introduction of that technique, clinicians routinely acid-etched, rinsed, and dried cavity preparations prior to bonding. What was not recognized at that time was that while normal mineralized dentin could be dried without shrinking, acid-etched dentin could not be dried without a two-thirds reduction in volume.14 This is because the stiffness of mineralized dentin is 19,000 MPa, while the stiffness of acid-etched dentin matrix is 1 MPa. The acid-etched dentin matrix is literally floating in the rinsing water. If that water is evaporated by air drying, the collagen fibril network collapses into a relatively impermeable organic film that interferes with resin infiltration15 (Figure 2c). Resin tags could still be formed and did provide some resin retention, but hybridization of the dentin between tubules (ie, intertubular dentin) could not occur. This left naked collagen fibrils in the acid-etched layer that could slowly hydrolyze and result in gap formation between resin and dentin. Kanca discovered that if some residual water was left in acid-etched dentin, bond strengths could be doubled.9-11
 |
| Figures 4a and 4b. Schematic (a) and transmission electron micrograph (b) of the “over-wet” phenomenon. In the total etch, wet bonding technique, there is the danger that some regions in complex cavity preparations (such as proximal boxes) may be too wet. When single bottle primer/ adhesives are applied, the solvent may diffuse into the water, instead of vice versa, forcing adhesive monomers to undergo phase changes, resulting in blisters and resin globules. Note that although the etched intertubular dentin has taken up resin to form hybrid layers (red zone), the adhesive (blue) did not form resin tags, but left the tubules in communication with microblisters filled with water (light blue). These blisters may compress when the restoration is under occlusal function, forcing dentinal fluid toward the pulp and causing postoperative sensitivity. This is one of the possible complications of this technique-sensitive bonding method. Courtesy of Dr. Franklin Tay, University of Hong Kong, with permission. |
Similar observations were made independently in Japan by Sugizaki.16 Careful scanning electron microscopy by Gwinnett17,18 revealed that wet bonding created thicker hybrid layers (ie, more resin uptake into etched surfaces) than did dry bonding. However, the application of acetone or ethanol as a means of removing water from the wet collagen fibril meshwork is only partially effective. The water often diffuses into the organic solvents so quickly that the monomers can no longer remain dissolved in the solvents. They undergo phase changes (Figure 4a and 4b) that can lead to low bond strengths because of the formation of resin globules19 and poor resin tag formation. This often results clinically in postoperative sensitivity.18
 |
 |
| Figures 5a and 5b. Use of so-called single-bottle, 2-step, total-etch, wet-bonding technique. The first step involves acid etching (a) to remove the smear layer/smear plugs and peritubular dentin. The second step is the application of the first layer of the combined primer/adhesive mixture (single bottle) that serves as a primer (b; light green in tubules), followed by a second layer (green) that serves as the adhesive (green surface layer). Modified from Bouillaguet10 with permission. |
Some manufacturers formulated dentin primers in acetone, while others used ethanol or water. More recently, the primer components have been mixed with adhesives in a single bottle to reduce the number of bonding steps. However, a separate acid-etching step is still required in these “total-etch” wet-bonding techniques. (Figure 5a) This is followed by application of 2 layers of bonding agent to wet dentin. The first layer serves the same purpose as did the original primer (purple layer, Figure 3b). It removes much of the residual water and begins to infiltrate adhesive monomers into the acid-etched dentin. If the acid-etched dentin surface is too wet, phase changes can occur. Generally, when the second layer of bonding agent is applied, the fresh monomers and their solvent re-dissolve the resin globules, leaving a more homogeneous film (green layer, Figure 5b).
It appears that the degree of surface wetness that is optimal for ideal dentin bonding varies widely among commercial products. There is no simple answer to the question of what is “too wet” or “too dry.” Thus, total-etch products are technique-sensitive. When used in the laboratory on flat dentin surfaces, one can obtain a relatively uniform surface “wetness.” Clinically, however, there is a tendency to over dry the pulpal or axial wall of complex cavities and to pool water at axial-gingival line angles, leaving the dentin surface with a nonuniform degree of wetness and nonuniform resin infiltration. The gingival floor of proximal boxes is often too wet to achieve perfect bonding, leading to microleakage and clinical sensitivity in those areas.
 |
| Figure 6a through 6c. Use of self-etching primer adhesive systems. In this dry-bonding technique, the smear layer serves as the substrate (a). An acidic primer is applied (light blue) that etches through the smear layer and into the top of the smear plugs in 20 seconds (b). After evaporating the solvent, an adhesive layer is applied (yellow) and light-cured to seal the surface (c). Modified from Bouillaguet10 with permission. |
To avoid the problems associated with total-etch/wet-bonding techniques, several groups began to explore a new approach to dentin bonding in the early 1990s. All adhesive formulations used in total-etch bonding systems contain 5% to 10% (by weight) of acidic monomers with other monomethacrylates or dimethacrylates. The pH of these formulations is between 2.5 and 4.5. This is not acidic enough to demineralize smear layers or etch dentin, hence the requirement for a separate step of acid etching with 32% to 37% phosphoric acid (pH 0.1 to 0.6). However, if the concentration of acidic monomers was increased from 5% to 10%, to 30% to 40% (by weight), and if these acidic monomers were dissolved in 30% to 40% (by weight) hydroxyethyl methacrylate (HEMA, a very water-soluble priming monomer), a new formulation was obtained that was both a self-etching and self-priming adhesive system with a pH low enough (ie, pH 1 to 2) to etch through smear layers (Figure 6a) into the underlying dentin (Figure 6b). The smear layers are either incorporated into the hybrid layer if the solution cannot be well agitated19,20 (ie, in distal proximal boxes), or they are completely dissolved if the surface can be scrubbed with the product. Importantly, the self-etched, self-primed surfaces are not to be rinsed with water. The surfaces are scrubbed for 20 seconds and then gently air-dried to evaporate the small amount of water that is in the product in order to ionize the acidic monomers and facilitate solubilization of calcium and phosphate ions from the etched smear layer and underlying intact tooth structure. The dried surface is then covered with a relatively hydrophobic adhesive layer that is light-cured (Figure 6c). The photoinitiators and accelerators in the primer ensure that the primer and adhesive cure together in a homogeneous fashion.
 |
| Figure 7. Schematic summarizing the use of self-etching primer adhesive systems. The self-etching primer (violet) is applied to dry, smear layer-covered enamel and dentin. After agitating for 20 seconds, the solvent is evaporated. There is no rinsing step. The primer dissolves or disperses the smear layer without removing all of the smear plugs. The primed dentin is then sealed with a layer of adhesive (purple) that is then light-cured. |
This new approach to bonding comes full circle to where dentin bonding began; ie, at the smear layer. Although the early adhesives were not acidic enough to etch through the smear layer and were too hydrophobic to adequately wet the smear layer, the new self-etching primers can utilize the smear layer as a legitimate bonding substrate (Figure 7). A great advantage is that self-etching primers are designed to be used on dry dentin. Although the dentin should not be desiccated, the dentin surface can be briefly dried following cavity preparation because the dentin is mineralized. This avoids questions regarding how moist or wet the dentin should be prior to bonding. It is far easier to produce uniform dryness than it is to produce uniform wetness. Another advantage of these self-etching primer systems is that they do not etch very far into the dentin beneath the smear layer. This avoids removal of smear plugs in the tubules (Figure 7) and accounts for the lack of postoperative sensitivity associated with these adhesive systems.20 The shallow etch ensures good resin infiltration. Even through the hybrid layer is thin,21 resin dentin bond strengths are very high.22
The first marketed modern self-etching primer (introduced in 1989) consisted of 2.5% maleic acid and 55% HEMA in water (pH 2).23 This self-etching, self-priming system had a relatively short shelf life, and the acidity of the maleic acid accelerated the breakdown to HEMA.
There was no evidence that the double-bond in maleic acid ever co-polymerized with HEMA. In 1992, a light-cured, self-etching primer was introduced. Its acidic monomer, Phenyl-P, did copolymerize with the other methacrylate comonomers. This was followed by the introduction of a dual-cure, self-etching primer system. Recent improvement in catalysts and adhesive monomers have taken place, and the introduction of a new antibacterial self etching primer system is expected in 2003.
Self-etching primer systems should not be confused with certain all-in-one self etching, self-priming, self-adhesive systems that are more acidic (pH < 1) than self-etching primers. These more acidic systems etch more deeply. They are also more hydrophilic and tend to produce adhesive films that are overly thin (5 to 10 µm). Air can diffuse into liquid monomers to a depth of 10 µm in seconds. When light-cured, the free radicals that are generated are consumed by oxygen in the adhesive, leading to a lack of polymerization of the monomers even after light curing. When composite filling materials are placed on these unpolymerized resins, they displace the adhesive monomers laterally, leaving resin composites in direct contact with hybrid layers. It is desirable to have a thicker (approximately 20 to 50 µm) layer of cured adhesive resin between resin composites and hybrid layers to avoid the problems associated with thin oxygen-inhibited layers. This layer of cured adhesive resin functions as a shock absorber.
 |
| Figures 8a and 8b. Transmission electron micrographs of dentin bonded with an all-in-1 adhesive, then placed in 37°C water for 24 hours prior to soaking in silver nitrate to demonstrate the presence of water-filled (now silver-filled) channels called “water trees” (pointers), extending from the hybrid layer (h) and passing through the cured adhesive layer. The black silver deposits indicate where there were water-filled channels. Such water trees do not form in self etching primer adhesives.23 Courtesy of Dr. Franklin Tay, University of Hong Kong, with permission. |
The second problem with all-in-one adhesives is that even if they are thick enough to polymerize, their composition is very acidic and very hydrophilic. That is, they attract and absorb water, leading to leaching of unpolymerized monomers or hydrolytic degradation products through water-filled channels called water trees24 (Figures 8a and 8b). Experimentally, the presence of water-filled voids and channels are disclosed by soaking bonded specimens in silver nitrate solutions. The silver nitrate diffuses into the water-filled spaces. Once reduced, black silver deposits are formed. These channels pass from the hybrid layers, through the adhesives, to the adhesive-composite interface. Adhesive systems that form water trees tend to have a lower bond strength to dental hard tissues, and are less durable than self-etching primer systems. In contrast, such water absorption does not occur in self-etching primer adhesive systems because the etched and primed dentin is sealed by a relatively thick, relatively hydrophobic adhesive layer that prevents water sorption.
CONCLUSION
Dentin bonding has evolved from no-etch to total-etch to self-etch systems. The development of self etching primer adhesive systems has greatly simplified resin bonding procedures, as a separate etching step is no longer required.25 In addition, a rinsing step is not required. Bonding can be accomplished under relatively dry conditions, avoiding the variables associated with wet bonding. These self-etching primer adhesives can be used to simultaneously etch both enamel and dentin.26 They bond equally well to superficial and deep dentin27 and can be used in place of liners and bases in deep cavities20 if they are covered with an intermediary resin composite. Furthermore, the use of caries disclosing agents has no effect on their bond strength.28
References
1. Kramer IRH, McLean JW. Alterations in the staining reactions of dentine resulting from a constituent of a new self-polymerizing resin. Brit Dent J. 1952; 92:150-153.
2. Davidson CL, deGee AJ, Feilzer AJ. The competition between composite dentin bond strength and the polymerization contraction stress. J Dent Res. 1984;63:1396-1399.
3. Buonocore MG. A simple method of increasing the adhesion of acrylic filling materiaJs to enamel surfaces. J Dent Res. 1955;34:849-853.
4. Buonocore MG, Wileman W, Brudevold F. A report on a resin composition capable of bonding to human dentin surfaces. J Dent Res. 1956;35:846-851.
5. Pashley DH, Ciucchi B, Sano H. Dentin as a bonding substrate. Dtsch Zahn Z. 1994;49:760-763.
6. Nakabayashi N, Kojima K, Masuhara E. The promotion of adhesion by the infiltration of monomers into tooth substances. J Biomed Mater Res. 1982;16:265-273.
7. Nakabayashi N, Pashley DH. Hybridization of Dental Hard Tissues. Chicago, Ill: Quintessence Publishing Co, Ltd; 1998.
8. Fusuyama T. New Concepts in Operative Dentistry. Chicago, Ill: Quintessence Publishing Co, Inc; 1980.
9. Pashley DH. The effects of acid etching on the pulpodentin complex. Oper Dent. 1992;17:229-242.
10. Bouillaguet S. Bonding resin-based materials to the dentin-pulp complex. [Thesis.] University of Geneva, Geneva, Switzerland, 2002:46.
11. Kanca J. A method for bonding to tooth structure using phosphoric acid as a dentine-enamel conditioner. Quintessence Int. 1991;22:285-290.
12. Kanca J. Improved bond strength through acid etching of dentin and bonding to wet dentin surfaces. J Am Dent Assoc. 1996a;123:35-43.
13. Kanca J. Wet bonding: Effect of drying time and distance. Am J Dent. 1996; 9:273-276.
14. Carvalho RM, Yoshiyama M, Pashley EL, Pashley DH. In vitro study on the dimensional changes of human dentine after demineralization. Arch Oral Biol. 1996;41:369-377.
15. Pashley D14, Ciucchi B, Sano H, Horner JA. Permeability of dentin to adhesive resins. Quintessence Int. 1993;24:618-63 1.
16. Sugizaki J. The effects of various primers on dentin adhesion of resin composites. Jpn T Conserv Dent. 1991;34:228-265,
17. Gwinnett AJ. Moist versus dry dentin: Its effect on shear bond strength. Am J Dent 1992; 5: 127-129.
18. Gwinnett AJ. Dentin bond strength after air drying and rewetting. Am J Dent. 1994;144-148.
19. Tay FR, Gwinnett AJ, Wei SHY. Micromorphological spectrum from overdrying to overwetting acid-conditioned dentin in water-free, acetone based, single-bottle primers/ adhesives. Dent Mater. 1996;12:236-244.
20. Unernori M, Matsuya Y, Akashi A, Goto Y, Akarnine A. Composite resin restoration and postoperative sensitivity: clinical follow-up in an undergraduate program. J Dent. 2001;29:7-12.
21. Watanabe L, Nakabayashi N, Pasbley DH. Bonding to ground dentin using a self-etcbing Phenyl-P primer. J Dent Res. 1994;73:1212-1220.
22. Tay FR, Carvalho R, Sano H, Pashley DH. Effect of smear layers on the bonding of a self-etching primer to dentin. J Adhes Dent. 2000;2:99-116.
23. Swift J. Bonding systems for restorative materials-a comprehensive review. Pediatric Dent. 1998;20:80-84.
24. Tay FR, Pashley DH. Water treeing-a potential mechanism for degradation of dentin adhesives. Am J Dent. 2002; (in press).
25. Christiansen G. Self-etching primers are here. J Am Dent Assoc. 2001;132:1041-1043.
26. Shono Y, Terashita M, Pashley EL, Brewer PD, Pashley DH. Effects of cross-sectional area on resin-enamel tensile bond strength. Dent Mater. 1997;13:290-296.
27. Pereira PNR, Okuda M, Sano H, Yoshikawa T, Burrow MF, Tagami J. Effect of intrinsic wetness and regional difference on dentin bond strength. Dent Mater. 1999;15:46-53.
28. Kazemi RB, Meiers JC, Peppers K. Effect of caries disclosing agents on bond strengths of total-etched and self-etching primer dentin bonding systems to resin composite. Oper Dent. 2002; 27: 238-242.
Dr. Pashley earned a DMD degree from the Oregon Health Sciences University in 1964 and then earned a PhD in physiology from the University of Rochester School of Medicine and Dentistry in 1970. He has held the positions of assistant professor, associate professor, and professor at the Medical College of Georgia. Currently, he is regents’ professor of oral biology in the School of Dentistry and professor of physiology and endocrinology in the School of Medicine at the Medical College of Georgia. Dr. Pashley’s research activities have been supported by the NIDR since 1973, and he has published over 300 papers in peer-reviewed journals about two thirds of which deal with the structure and function of dentin. He has held various offices in the Pulp Biology Group of the IADR and received the Pulp Biology Research Award (one of the FADR Distinguished Scientist Awards) in 1990 for his research and contributions to the field of pulp biology. He received the Hollenbach award from the Academy of Operative Dentistry in 1998, and, in 2001, he received the Wilmer Souder award for this work in Dental Materials. Dr. Pashley serves on the editorial boards of the Journal of Adhesive Dentistry, Dental Materials, the Journal of Endodontics, and the Journal of Dentistry. He lectures nationally and internationally and participates in international conferences in his area of research. He is a consultant to the American Dental Association and to a number of domestic and foreign dental products companies. He can be reached at (706) 721-2033.










