Single-Tooth and Extended Edentulous Restorations
BACKGROUND
Titanium and titanium alloy implants have been the materials of choice in the manufacturing of dental implants for more than 5 decades. They have been thoroughly investigated and have enjoyed great success thanks to improvements in macroscopic and nanoscopic surface improvements and design. Conversely, the popularity of metal alloy implants has come with increased reports ranging from poor aesthetics to immune reactions to these implants and their prosthetic components.1-3 Metal alloy orthopedic and dental implants are well documented to be able to induce hosts’ responses to their alloy components. The responses have been reported to range from local soft-tissue irritation to spontaneous unexplainable and often asymptomatic implant failure, joint pain, skin irritation, fatigue, osteolysis, and a host of other systemic health issues.
Furthermore, there is a significant body of research that has proven that titanium implants, when exposed to bodily fluids and submitted to functional cycles and the rigors of the oral environment, will undergo corrosion attacks and release metal ions and particles in the peri-implant tissues and distant organs.4-6 In a recent study, Daubert et al7 were able to establish the connection between the release of metal ions, increased inflammatory mediators activity, and peri-implantitis. Now that research on, and the use of, load-bearing bioceramics has matured and is growing exponentially in medicine and dentistry, there is a paradigm shift in the types of materials used for implantation and restoration of dental implants. Bioceramics and other bioinert, biocompatible, non-metal materials are rapidly replacing metal alloys. Yttria-stabilized tetragonal zirconia polycrystal, and zirconia composites such as zirconia-toughened alumina and alumina-toughened zirconia (ATZ) are fast becoming the materials of choice for dental implantation and fixed prosthetics.8 These polycrystal zirconia composites, thanks to their superior biomechanical properties;9 the absence of galvanic activity, low plaque and bacterial accumulation,10 and resistance to oxidation, are rapidly establishing themselves as the implantable bioceramics of choice.
The goal of this series of articles is to highlight the features of ceramic implants, their advantages, and their direct clinical applications in a broad range of cases. Part 1 of this series will discuss single-unit and multi-tooth edentulism treatment with ceramic implants. Part 2 will focus on full-arch, removable solutions with ceramic implants.
CASE REPORTS
Case 1: Congenitally Missing Upper Premolar
A 37-year-old, healthy female patient presented to our clinic for a tooth-replacement consultation. She had been missing her upper left first premolar congenitally and had a cantilever bridge off of the second premolar, which had clinically detectable open margins and recurrent decay (Figure 1).
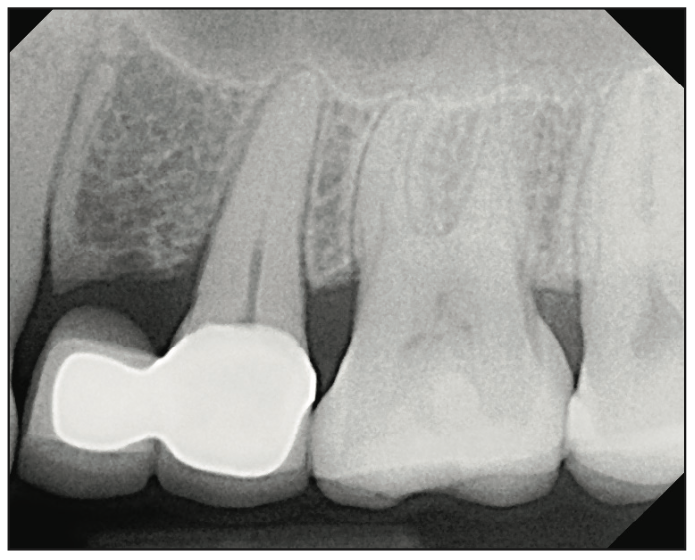
Figure 1. Periapical radiograph of the edentulous area showing a cantilever bridge from Nos. 12 to 13.
Replacement options were discussed, and the patient chose a fixed replacement solution and requested a metal-free implant. One of the reasons for choosing a ceramic implant was that the patient was a dental hygienist, and she had been impressed with the health of soft tissues around these implants on the patients she treated. Local anesthesia was administered by infiltration on the buccal and palatal sides, implant osteotomy was done using copious irrigation and manufacturer protocols, and a 4.2- × 10-mm Zeramex XT implant (Dentalpoint AG) was placed (Figure 2). The final torque of the implant placement was 35 N/cm2; the implant stability quotient (ISQ) values of 67 were measured (Figure 3) using the Osstell Beacon device (W&H Dentalwerk). A healing abutment was placed onto the implant (Figure 4), and a provisional cantilever bridge was made during the healing period.
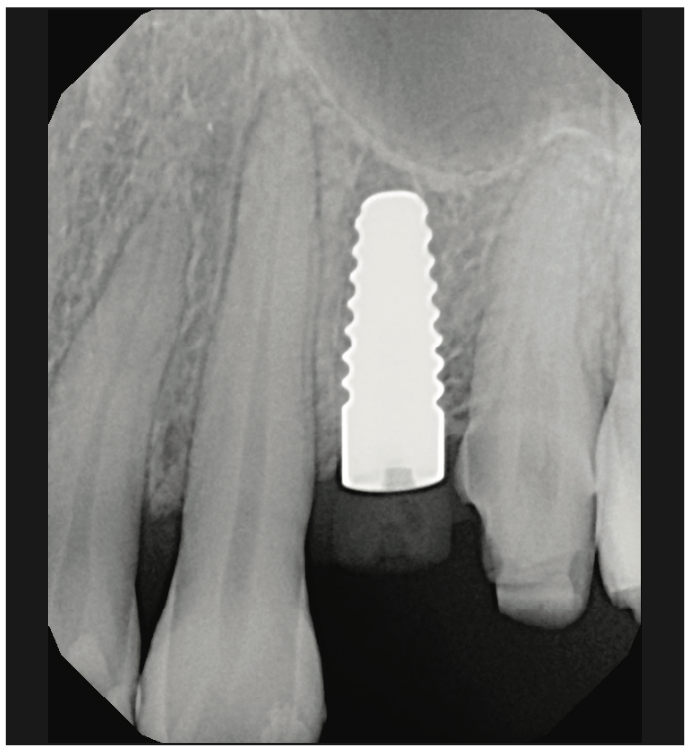
Figure 2. Final periapical radiograph of a 4.2- × 10-mm Zeramex XT ceramic dental implant (Dentalpoint AG) placed at site No. 12.
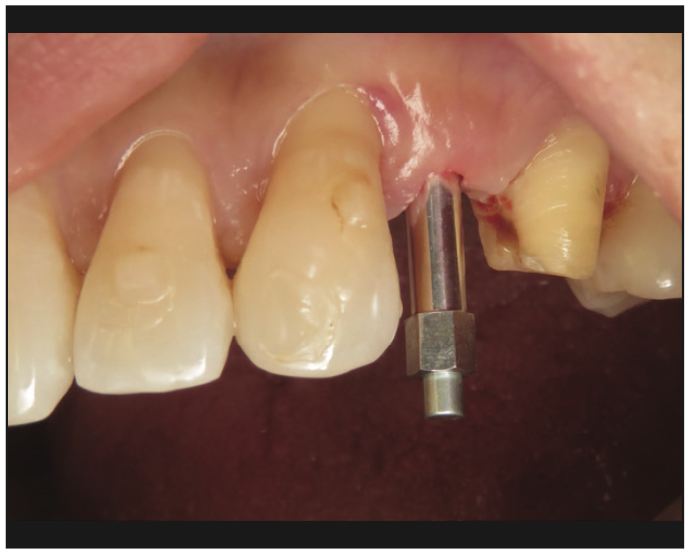
Figure 3. Clinical photo showing the Osstell SmartPeg positioned over the implant to measure implant stability.
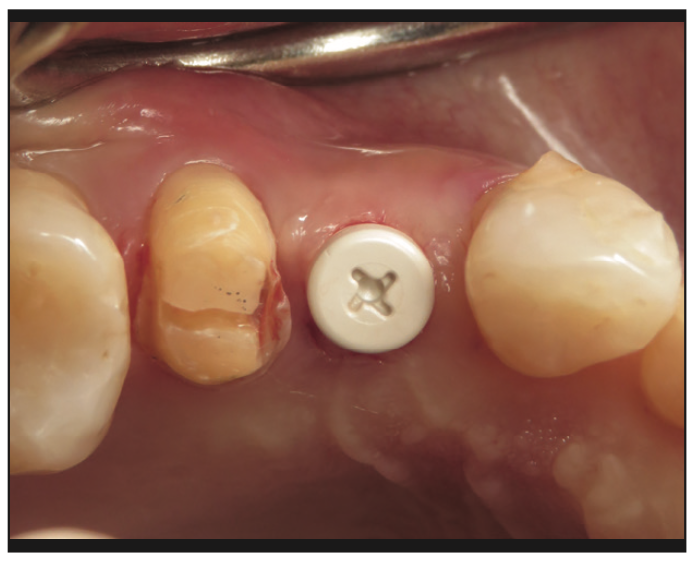
Figure 4. Clinical photograph showing the healing abutment immediately after a minimally invasive implant placement.
Four months after the implant placement, the patient returned for the restorative phase. Clinical examination showed healthy soft tissues around the implant with an abundance of keratinized gingivae. A closed-tray impression coping (Figure 5) was used for the final impression; a bite registration and photographs were also captured to allow for the fabrication of the final prosthesis. The tooth was restored with a screw-retained, layered zirconia crown over a customized zirconia abutment. Radiographs were used to confirm the seat of both the abutment and crown (Figure 6). The final photo shows natural aesthetics and harmony of the restoration blending with the adjacent tissues (Figure 7).
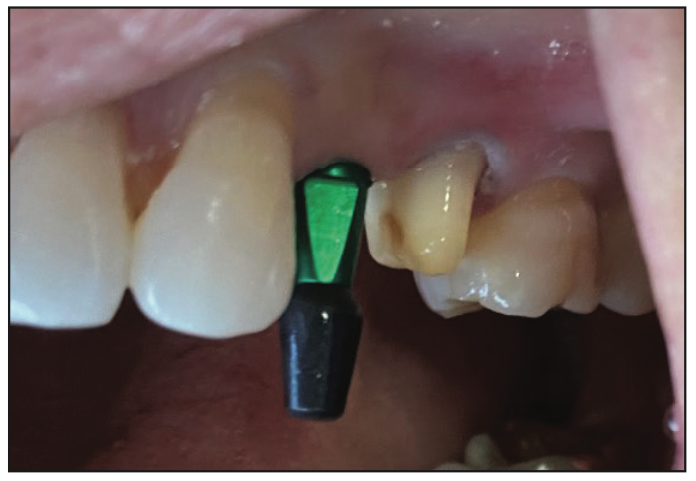
Figure 5. A closed-tray impression coping was used to capture implant position and surrounding structures.
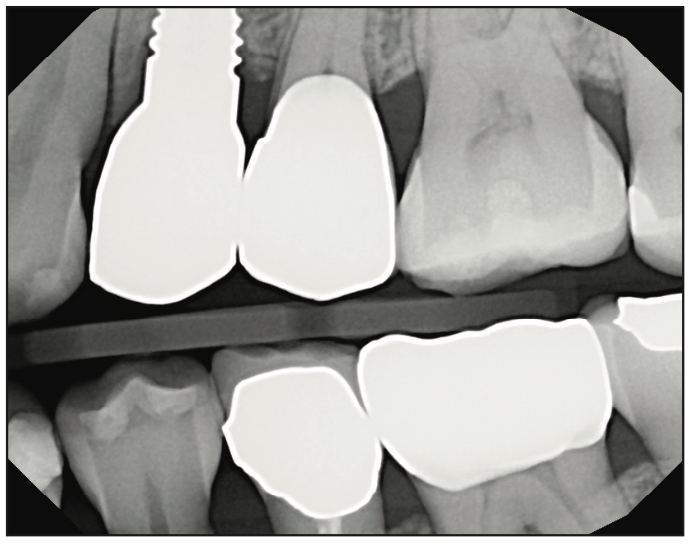
Figure 6. Final bite-wing radiograph showing complete seat of the restoration on implant No. 12 and a new crown on No. 13.
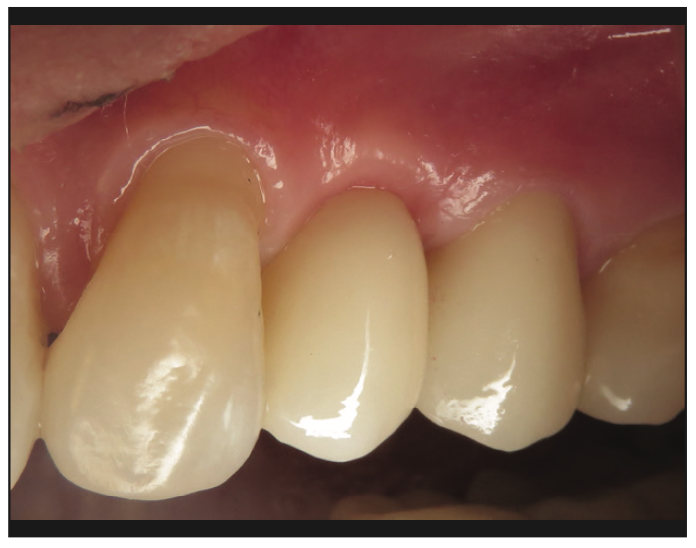
Figure 7. Clinical photo of the final restorations showing perfect blending of the implant restorations and healthy gingival tissues.
Case 2: Mandibular Aesthetic Zone Implants
An ASA I, 53-year-old male patient presented to our clinic for a post-orthodontic treatment consultation to replace tooth No. 26. The patient, for lifestyle and personal reasons, elected to replace his missing anterior mandibular incisor and the adjacent failing central incisor with metal-free implants and non-metallic prosthetic materials and components. Upon clinical and radiographic examination, tooth No. 25 had root resorption and Type II to III mobility (Figure 8). Clinical examination and CBCT showed adequate bone (Figure 9) and soft tissue in both sites and favorable conditions for implant placement at Nos. 26 and 25. The final treatment plan included extraction of tooth No. 25 with immediate implant placement and simultaneous placement of implant No. 26 in a healed site. There was, however, limited space, and it was decided to use 2 narrow-diameter ceramic implants. The surgery was done after administering local anesthesia by infiltration technique for a total of 4 carpules of Lidocaine 2% with 1:100,000 epinephrine. Tooth No. 25 was extracted atraumatically, and the socket was curetted and then rinsed with saline.
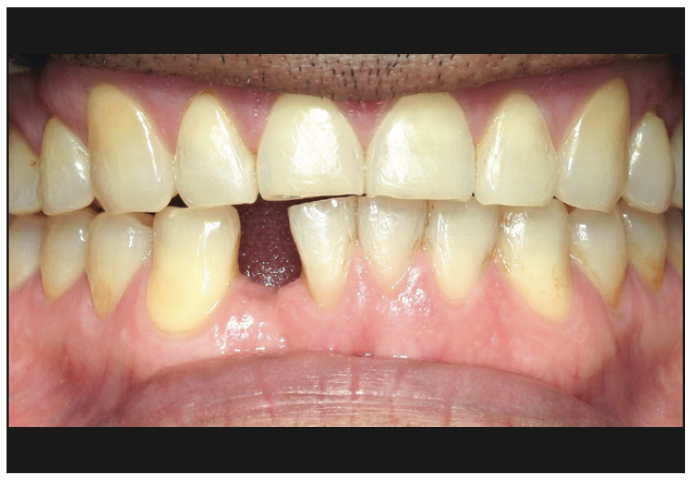
Figure 8. Preoperative photograph of the mandibular anterior area.
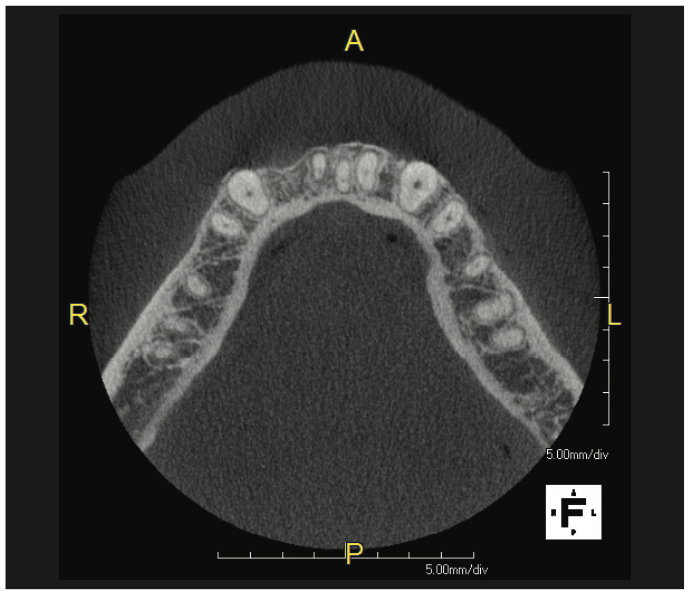
Figure 9. Pre-op CBCT showing adequate bone volume at the No. 26 site and a thin buccal plate on the No. 25 site.
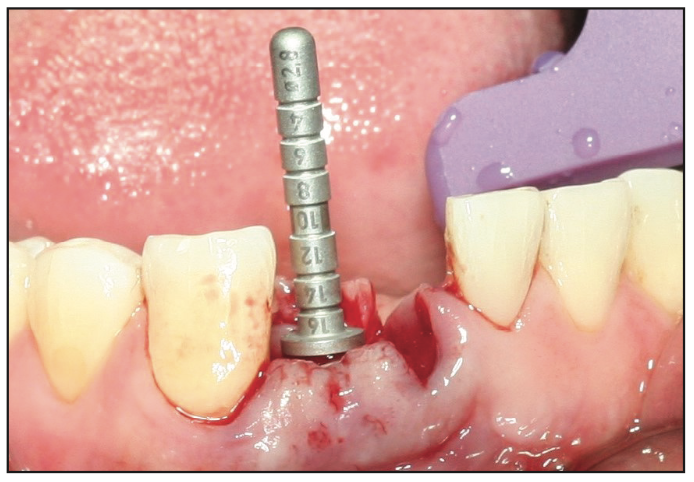
Figure 10. Intra-operative clinical photo showing the use of the direction indicator for depth and angulation control.
Given the limited mesio-distal space available, the manufacturer’s osteotomy protocol was slightly modified by using osseocondensation Densah drills (Versah USA) in the delayed placement of implant No. 26 (Figure 10). The osteotomy for No. 25 was prepared according to the manufacturer’s protocol. Two 3.3- × 10-mm Zeramex P6 ceramic implants (Dentalpoint AG) were inserted (Figure 11). The final insertion torque was 35 N/cm2 for both implants, and using the Osstell Beacon the initial ISQ values of 79 and 86 were obtained for Nos. 25 and 26, respectively. Peek cover screws were placed, and the flaps were approximated using a 4-0 chromic gut suture. During the healing period, the patient wore a Hawley retainer with acrylic teeth as an aesthetic provisional. The 8-week follow-up appointment showed excellent healing and healthy gingival tissues around the implants (Figure 12).
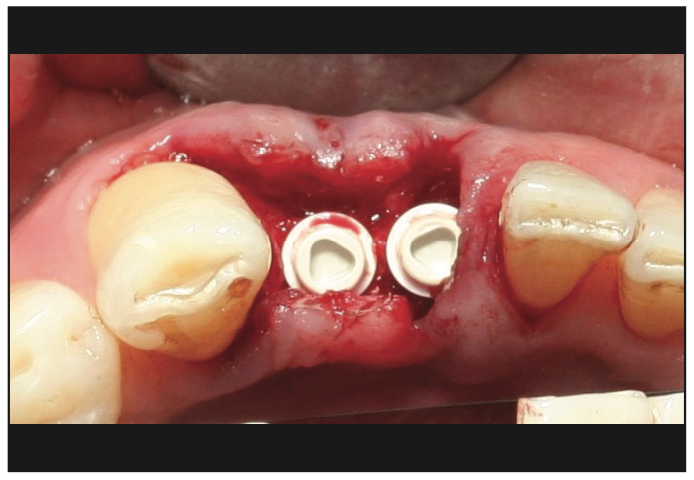
Figure 11. Final placement of both implants in a prosthetically referenced position.
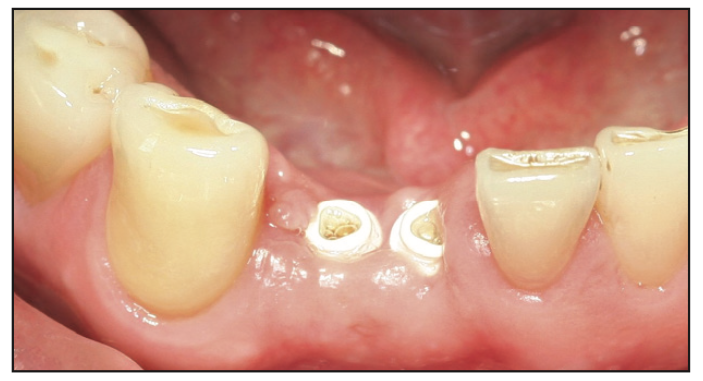
Figure 12. Excellent healing of the tissues at 8 weeks after surgery.
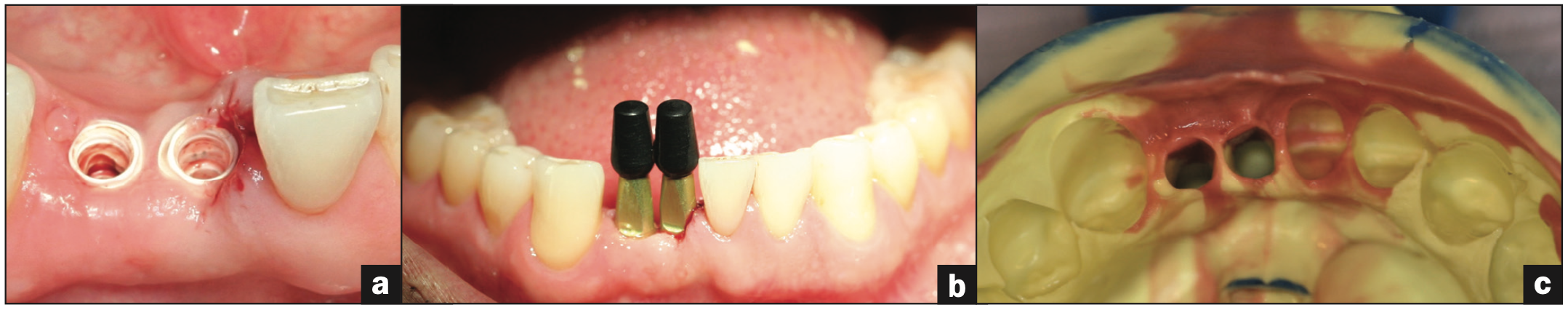
Figure 13. (a) Excess soft tissue trimmed to allow for impression coping seating. (b) Closed-tray impression copings seated onto the implants. (c) Final impression with heavy- and soft-body PVS materials.
After 4 months of healing time, stage II procedures were initiated. Closed-tray impression copings were used to capture the position of the implants and adjacent structures (Figure 13). The patient wore provisional crowns for 2 to 3 months (Figure 14), and after that, final, all-ceramic restorations were made over customized, prefabricated zirconia abutments. The abutments, just like the implants, were made of ATZ and were customized in the dental laboratory. They were connected to the implants using the Vicarbo prosthetic screw, which is metal-free and made from medical-grade carbon fiber-reinforced peek material. The crowns were delivered successfully. Final clinical photos and radiographs (Figures 15 and 16) show the ideal placement of the implants in the aesthetic zone and harmonious restorations that blend with the existing natural dentition and gingivae.
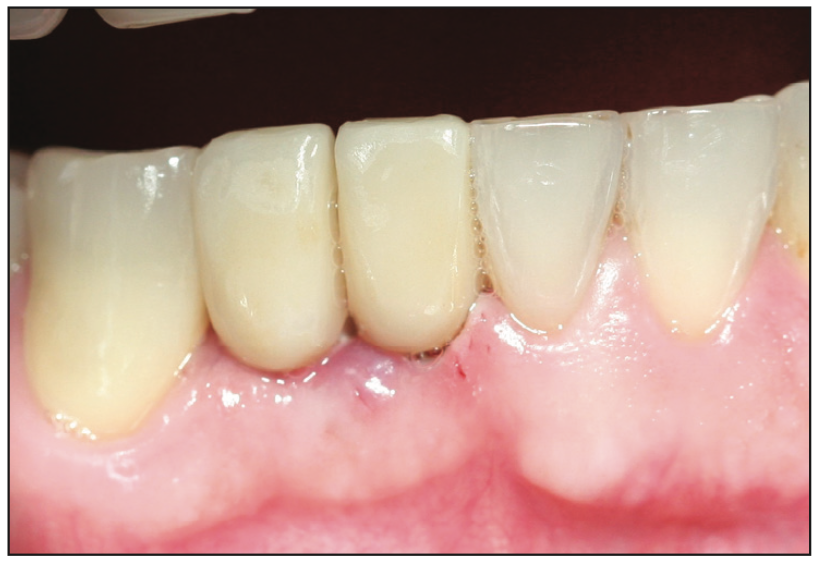
Figure 14. Clinical photo of the provisional crowns.
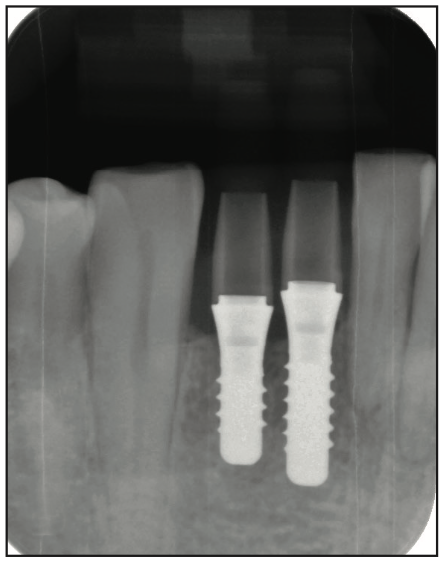
Figure 15. Periapical radiograph showing the implants in ideal position in bone and parallel to each other.
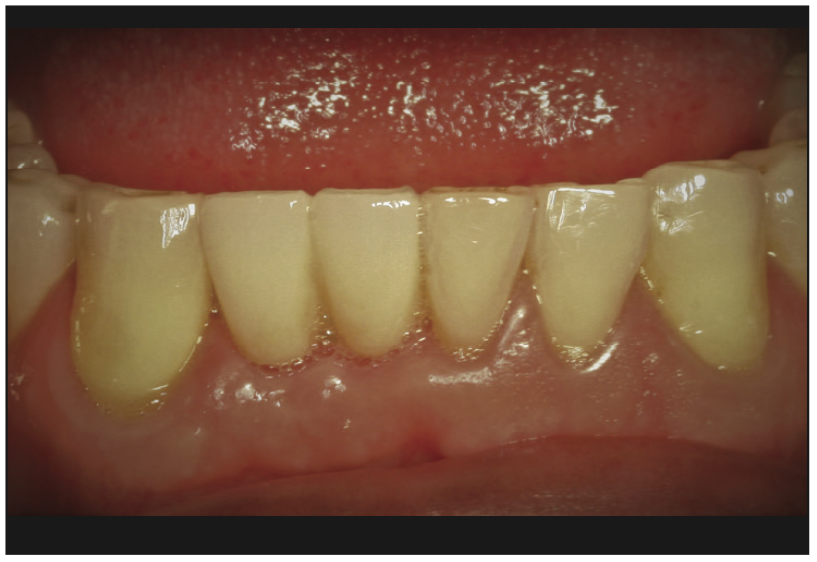
Figure 16. Final clinical photograph of the restorations that blend in harmoniously with surrounding tissues and dentition and healthy gingival tissues.
DISCUSSION
Ceramic dental implants are becoming more and more popular due to a demand for highly biological, biocompatible, aesthetic, and metal-free solutions for the replacement of missing teeth. What makes ceramic implants desirable for patients is their aesthetic and biocompatibility advantages over their titanium counterparts (no corrosion and no dark hue of the gums due to titanium) and their bio-inert and biological properties. For the clinician, the advantages go deeper than the surface and aesthetic value—zirconia exhibits less plaque accumulation; comparable and, lately, faster osseointegration and higher bone-to-implant contact percentages; and proven superior aesthetics. In comparison to titanium, ceramic implants are not subjected or vulnerable to corrosion attacks; therefore, unlike metal implants, they do not release titanium, aluminum, and other alloy component ions into the surrounding tissue through a process called tribocorrosion. Tribocorrosion, and the resulting release of titanium ions, contribute to the increased levels of inflammation in the soft and hard tissues. The inflammatory mediators contribute to an increase in osteoclastic activity and osteolysis and are shown to contribute to and play a significant role in peri-implant bone loss and peri-implantitis.11,12
Many years of interest and research in ceramic dental implants have resulted in the advancement of both the chemical composition of the zirconia and the manufacturing of these implants. Given the material instability when exposed to humidity, commercially available ceramic implants are not made with 100% zirconia; the material is reinforced with yttria, ceria, or different levels of alumina, which improves the physical properties of the implants, their aesthetic value and stability at body temperature, and make them more suitable for long-term function and harmony in the oral cavity. The new zirconia implants are stronger, with greater flexural strength, and less susceptible to cracks and crack propagation. These advanced properties give the clinician, in the case of one-piece implants, the ability to prepare the cervical section of the implant itself to improve prosthetic clinical outcomes. The advances in manufacturing techniques (injection molding, 3D printing, and hot isostatic pressing) have allowed for the creation of a wide variety of load-bearing bioceramics, including one-piece and 2-piece configurations, both as bone and as tissue-level implants. The evolution of manufacturing techniques and protocols have led to material advances that have made it possible to have narrow-diameter zirconia implants as small as 3.3 mm, like the ones used in case 2. Microscopically and—lately—nanoscopically, there are various surface treatment options as well, namely sandblasting, laser etching, acid etching, photofunctionalization, and combinations thereof that improve osseointegration and provide surfaces favorable to effective mucointegration.13,14 The implants used in the cases presented had their subcrestal surfaces treated with a combination of sandblasting and acid etching. Given all these advancements and the wide variety of treatment solutions, it is no surprise that virtually all dental edentulous needs can be solved with ceramic implants. The second part of this article will present the use of ceramic implants in full-arch removable and fixed reconstructions.
CONCLUSION
The top 3 implant manufacturers in the world have ceramic implants in their portfolios, which, in North America, they are still timidly offering to their customers. Ceramic dental implants offer an excellent level of predictability and flexibility for both surgical and restorative clinicians and provide metal-free, highly biocompatible, aesthetic solutions for single-unit replacements or in extended edentulous sites. The evolution in mechanical properties and the aesthetic value of load-bearing bioceramics has improved and continues to improve significantly, so much so that, in this particular case, there were small-diameter ceramic implants available to replace 2 consecutive mandibular anterior incisors. The demand for ceramic implants and their rapid growth still remains driven, for the most part, by the patients, whom the authors regard as the secondary consumers of dental implants after the clinicians. After 2 decades of experience with ceramic implants between them, the authors have observed a faster and greater demand for ceramic implants from the public. However, in the last 3 years, thanks to continually increasing patient requests, improved macroscopic and microscopic designs, longer-term clinical reports, and greater availability of training opportunities in the United States, there has been a perceptible rise in interest from dentists in ceramic implants.
REFERENCES
1. Evrard L, Waroquier D, Parent D. Les allergies aux métaux dentaires. Un allergène émergent: le titane [Allergies to dental metals. Titanium: a new allergen]. Rev Med Brux. 2010;31(1):44–9. French.
2. Siddiqi A, Payne AGT, De Silva RK, et al. Titanium allergy: could it affect dental implant integration? Clin Oral Implants Res. 2011;22(7):673–80. doi:10.1111/j.1600-0501.2010.02081.x
3. Chaturvedi TP. An overview of the corrosion aspect of dental implants (titanium and its alloys). Indian J Dent Res. 2009;20(1):91–8. doi:10.4103/0970-9290.49068
4. Noumbissi S, Scarano A, Gupta S. A literature review study on atomic ions dissolution of titanium and its alloys in implant dentistry. Materials (Basel). 2019;12(3):368. doi:10.3390/ma12030368
5. Weingart D, Steinemann S, Schilli W, et al. Titanium deposition in regional lymph nodes after insertion of titanium screw implants in maxillofacial region. Int J Oral Maxillofac Surg. 1994;23(6 Pt 2):450–2. doi:10.1016/s0901-5027(05)80045-1
6. Lee S, Goh BT, Lai SH, et al. Peri-implant and systemic release of metallic elements following insertion of a mandibular modular endoprosthesis in Macaca fascicularis. Acta Biomater. 2009;5(9):3640–6. doi:10.1016/j.actbio.2009.05.028
7. Daubert D, Pozhitkov A, McLean J, et al. Titanium as a modifier of the peri-implant microbiome structure. Clin Implant Dent Relat Res. 2018;20(6):945–53. doi:10.1111/cid.12676
8. Piconi C, Maccauro G. Zirconia as a ceramic biomaterial. Biomaterials. 1999;20(1):1-25. doi:10.1016/s0142-9612(98)00010-6
9. Anders S. Zirconia as a biomaterial for odontological applications Effects of composition and manufacturing processes on fracture resistance. [Doctoral thesis] Umeå, Sweden: Umeå University; 2010.
10. Rimondini L, Cerroni L, Carrassi A, et al. Bacterial colonization of zirconia ceramic surfaces: an in vitro and in vivo study. Int J Oral Maxillofac Implants. 2002;17(6):793–8.
11. Yao Y, Cai X, Ren F, et al. The macrophage-osteoclast axis in osteoimmunity and osteo-related diseases. Front Immunol. 2021;12:664871. doi:10.3389/fimmu.2021.664871
12. Eger M, Hiram-Bab S, Liron T, et al. Mechanism and prevention of titanium particle-induced inflammation and osteolysis. Front Immunol. 2018;9:2963. doi:10.3389/fimmu.2018.02963
13. Dethier F, Bacevic M, Lecloux G, et al. The effects of abutment materials on peri-implant soft tissue integration: a study in minipigs. J Prosthodont. 2022;31(7):585–92. doi:10.1111/jopr.13504
14. Razali M, Ngeow WC, Omar RA, et al. An in-vitro analysis of peri-implant mucosal seal following photofunctionalization of zirconia abutment materials. Biomedicines. 2021;9(1):78. doi:10.3390/biomedicines9010078
ABOUT THE AUTHORS
Dr. Noumbissi obtained his DDS degree from Howard University in Washington, DC. He attended the prestigious Loma Linda University full-time graduate program in implant dentistry. There, he received 3 years of formal training in dental implantology, which culminated with a certificate in implant dentistry and an MS degree in implant surgery. He is a clinical and experimental researcher and author and has published book chapters and articles in implant dentistry with a focus on ceramic implants in peer-reviewed dental journals. He is a visiting professor at the University of Milan, an adjunct professor at the University of Chiety-Pescara in Italy, and a visiting researcher in the ceramic materials department at INSA Lyon in France. Dr. Noumbissi is the founder and current president of the International Academy of Ceramic Implantology and a Fellow and an ambassador at the Clean Implant Foundation. Dr. Noumbissi has extensive experience with ceramic implants, and his opinions and expertise are often sought both by clinicians and manufacturers. His practice is located in Silver Spring, Md. He can be reached at sammy@iaoci.com.
Disclosure: Dr. Noumbissi reports no disclosures.
Dr. Boyer graduated from the University of California, Los Angeles (UCLA) School of Dentistry in 2008. He completed 2 hospital-based GPR training programs at Cedars-Sinai Medical Center in Los Angeles and in the VA Healthcare system, where he received advanced training in oral and maxillofacial surgery, complex and comprehensive treatment planning, and the placement and restoration of dental implants. He has been in private practice in Los Angeles since 2010.Dr. Boyer is a faculty member at the UCLA School of Dentistry, where he teaches dental students as well as the residents in the Advanced Education in General Dentistry program. In 2020, he was invited to be one of 4 investigators in a 5-year, worldwide multicenter ceramic implant osseointegration and stability study led by the International Academy of Ceramic Implantology (IAOCI) and the Zirconia Implant Research Group. Dr. Boyer is active in multiple dental implant organizations and was recently appointed as a board member of the IAOCI. He can be reached at dentist@ucla.edu.
Disclosure: Dr. Boyer has previously lectured for Ostell. He did not recieve any financial compensation for writing this article.











