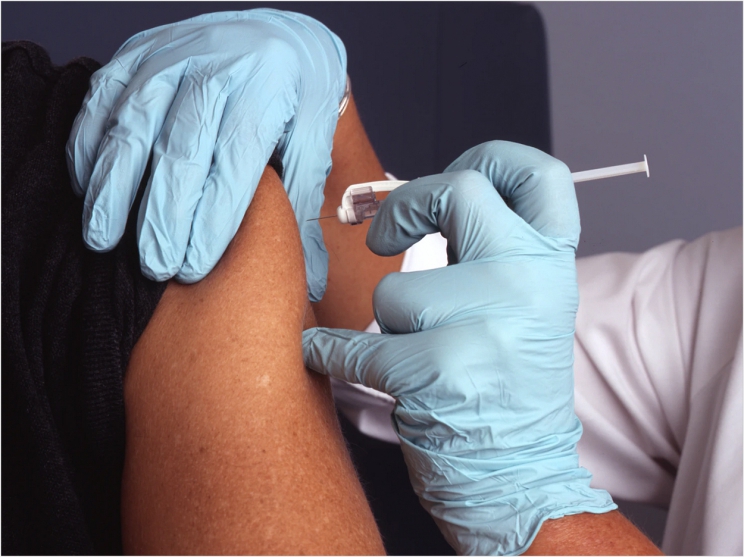
The State of California Department of Consumer Affairs is now allowing dentists to administer COVID-19 vaccines that the US Food and Drug Administration (FDA) has approved or authorized to people age 16 and older.
Dentists also may administer epinephrine or diphenhydramine by injection to patients who experience a severe allergic reaction as a result of the vaccine.
Before they can begin administering the vaccine, though, dentists must successfully complete training programs available through the Centers for Disease Control and Prevention, including:
- COVID-19 Vaccine Training: General Overview of Immunization Best Practices for Healthcare Workers
- What Every Clinician Should Know About COVID-19 Vaccine Safety
- What Clinicians Need to Know About the Pfizer-BioNTech and Moderna COVID-19 Vaccines
- Pfizer-BioNTech COVID-19 Vaccine: What Healthcare Professionals Need to Know
Additionally, dentists must comply with all applicable federal and state record-keeping and reporting requirements, including providing documentation to the patient’s primary care provider as applicable and entering information in the appropriate immunization registry designated by the immunization branch of the State Department of Public Health.
The subject vaccine must be administered in accordance with any applicable FDA emergency use authorization as well.
Related Articles
Ortho Resident Administers COVID-19 Vaccines
Dentists Will Be Essential in Providing COVID-19 Vaccinations
MDS Says Its Dentists Are Ready to Administer the COVID-19 Vaccine











