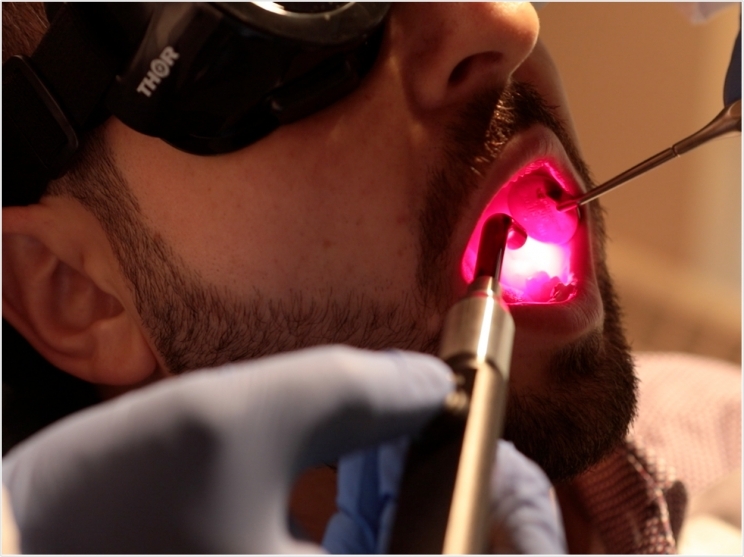
Photobiomodulation therapy is one of the most effective interventions for preventing the oral mucositis that often results from cancer treatment, according to researchers at the Eastman Institute for Oral Health. New and updated guidelines will help healthcare professionals deliver better care to these patients, the researchers added.
Frequently manifesting as painful sores and ulcers, oral mucositis is often reported by patients to be one of the worst side effects of cancer treatment. Pain from the condition can slow or delay cancer treatment, and in severe cases, it can require parenteral feeding and hospitalization.
Light therapies have existed for decades, but technology improvements have made them more affordable for wider use and provided more evidence proving their effectiveness. Low-level laser therapy has been proved to relieve pain or inflammation and promote healing, for example.
Photobiomodulation is considered routine therapy for oral mucositis across Europe, Brazil, India, and other nations. As the body of literature increases, the researchers said, it likely will be identified as beneficial for additional oral conditions.
“But even with the best evidence-based interventions, we don’t yet have an ultimate guideline for mucositis in all clinical settings,” said Sharon Elad, DMD, MSc, professor at the Eastman Institute for Oral Health and chair of its Oral Medicine Department.
“We look forward to future research to help shape a more universal implementation of photobiomodulation therapy as well as identify additional effective and validated protocols,” said Elad.
Elad also is chair of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) Mucositis Study Group (MSG), which aims to improve the outcomes of patients experiencing oral and gastrointestinal mucositis associated with cancer therapies. The group includes 245 members from 33 countries.
The researchers said that future studies are needed to verify the effectiveness of light therapy in managing oral mucositis in pediatric cancer patients and in adult cancer patients receiving only chemotherapy.
The MASCC/ISOO clinical practice guidelines covered a series of interventions categorized as:
- Anti-inflammatory agents
- Antimicrobials, coating agents, anesthetics, and analgesics
- Basic oral care
- Cryotherapy
- Growth factors and cytokines
- Laser and light therapy, also known as photobiomodulation therapy
- Natural therapies and miscellaneous
- Interventions for gastrointestinal mucositis
These clinical practice guidelines are evidence-based and were developed through meticulous and strict methods, according to the researchers. Also, the MSG conducted a systematic review about the pathogenesis of mucositis.
The first set of guidelines was published by Supportive Care in Cancer.
Related Articles
Researcher Uses Lasers to Regenerate Dental Tissue
Guidelines Recommend Light Therapy for Oral Mucositis Treatment
Grant to Support Research into Light Therapy for Treating Oral Pain


