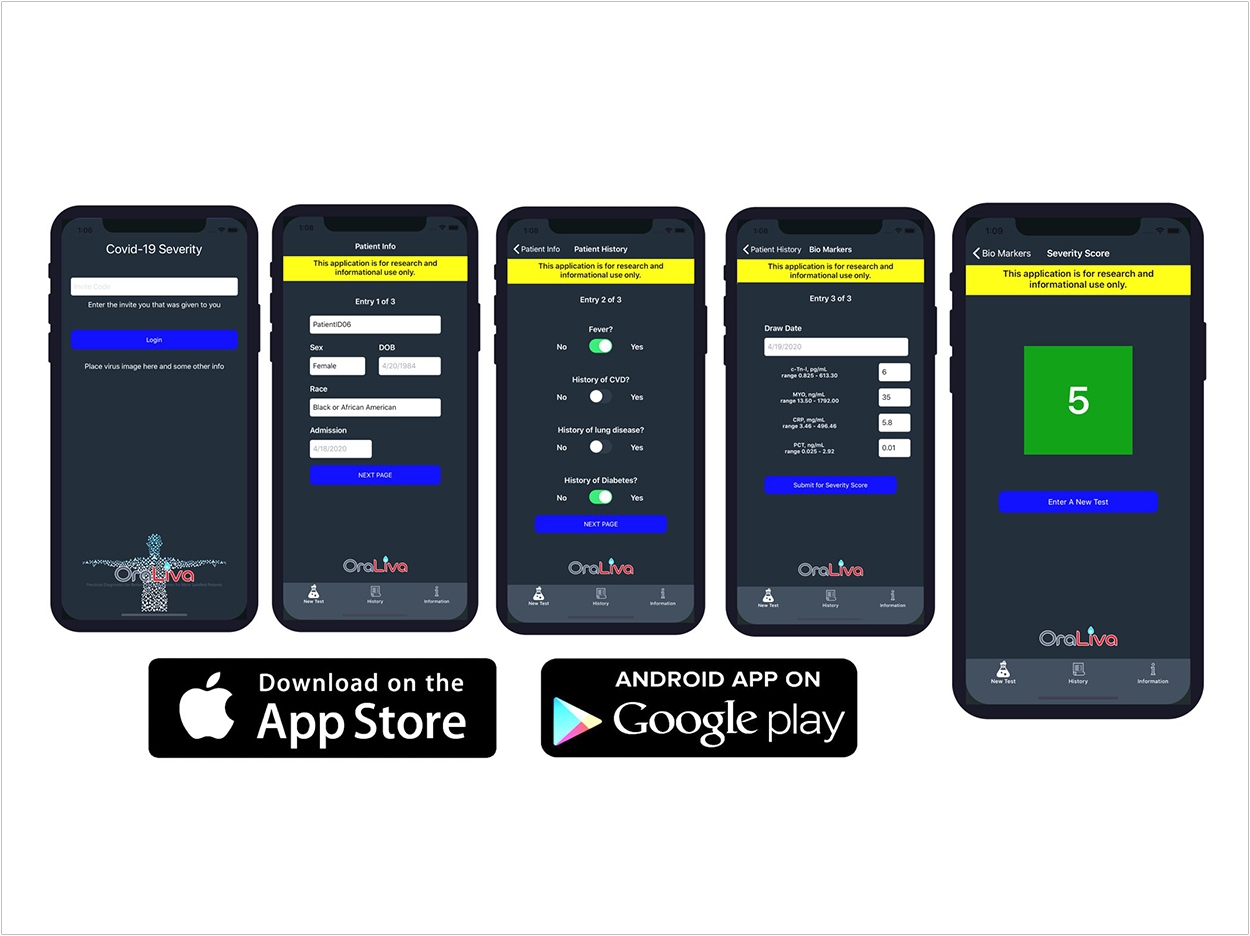
A mobile app created by researchers at the New York University (NYU) College of Dentistry can help clinicians determine which COVID-19 patients are likely to have severe cases. It uses artificial intelligence to assess risk factors and key biomarkers from blood tests to produce a “severity score.”
Current diagnostic tests for COVID-19 detect viral RNA to determine whether someone does or doesn’t have the virus, but they don’t provide clues as to how sick a COVID-positive patient may become, NYU said.
“Identifying and monitoring those at risk for severe cases could help hospitals prioritize care and allocate resources like ICU beds and ventilators. Likewise, knowing who is at low risk for complications could help reduce hospital admissions while these patients are safely managed at home,” said research leader John T. McDevitt, PhD, professor of biomaterials.
“We want doctors to have both the information they need and the infrastructure required to save lives. COVID-19 has challenged both of these key areas,” said McDevitt.
Using data from 160 hospitalized COVID-19 patients in Wuhan, China, the researchers identified four biomarkers measured in blood tests that were significantly elevated in patients who died versus those who recovered: C-reactive protein (CRP), myoglobin (MYO), procalcitonin (PCT), and cardiac troponin I (cTnI).
These biomarkers can signal complications that are relevant to COVID-19, NYU said, including acute inflammation, lower respiratory tract infection, and poor cardiovascular health.
The researchers then built a model using the biomarkers as well as age and sex, two established risk factors. They trained the model using a machine learning algorithm to define the patterns of COVID-19 disease and predict its severity.
When a patient’s biomarkers and risk factors are entered into the model, it produces a numerical COVID-19 severity score ranging from 0 (mild or moderate) to 100 (critical).
The model was validated using data from 12 hospitalized COVID-19 patients from Shenzhen, China, which confirmed that its severity scores were significantly higher for the patients who died versus those who were discharged.
As New York City emerged as the epicenter of the epidemic, the researchers further validated the model using data from more than a thousand patients in the city. To make the tool available and convenient for clinicians, the researchers developed a mobile app that can be used at point-of-care to quickly calculate a patient’s severity score.
The app has been retrospectively evaluated in the Family Health Centers at NYU Langone in Brooklyn, which serve more than 102,000 patients each year as one of the nation’s largest Federally Qualified Health Center networks, NYU said.
“Real-time clinical decision support tools for COVID-19 can be extremely helpful, particularly in the outpatient setting, to help guide monitoring and treatment plans for those at risk,” said coauthor Isaac P. Dapkins, MD, chief medical officer for the Family Health Centers at NYU Langone.
After optimizing the clinical utility of the app at the Family Health Centers in May, the researchers aim to roll it out nationwide in the coming weeks. It is possible that the COVID-19 severity score could be integrated into electronic health records, providing clinicians with actionable information at an early stage for those diagnosed with COVID-19, NYU said.
“We hope this tool can help identify those at high risk for adverse outcomes and reduce the health disparities present with COVID-19,” said Larry K. McReynolds, executive director for the Family Health Centers.
The COVID-19 severity score leverages a model McDevitt previously developed to predict outcomes for patients with cardiac disease. Cardiac health is one of several priorities of his lab, which creates point-of-care diagnostic systems that can be programmed to test for oral cancer, cardiac disease, and now COVID-19 biomarkers.
The diagnostic system uses small, noninvasive samples such as swabs of saliva or drops of blood from a fingertip added to credit card-sized cartridges armed with bio-nano chips pioneered by McDevitt. The cartridge is inserted into a portable analyzer that simultaneously tests for a range of biomarkers with results available in less than half an hour.
Because the technology is currently used for research and informational purposes only, the app can be used with existing laboratory tests and requires oversight by an authorized clinicians. But over the next few months, McDevitt’s laboratory, in partnership with SensoDx, a company spun out of his lab, plans to develop and scale the ability to test a drop of blood for COVID-19 severity biomarkers and produce a severity score on the spot.
“With COVID-19, point-of-care testing, coupled with a decision support system, could improve how clinicians triage patients and potentially improve their outcomes, particularly for those who need more immediate and aggressive care,” McDevitt said.
In addition to McDevitt’s research group at NYU, the study involved collaborators from NYU Grossman School of Medicine, NYU Tandon School of Engineering, Zhongnan Hospital of Wuhan University, and Lathan BioPharm Group.
The app was developed by McDevitt’s laboratory and OraLiva, a company founded by McDevitt. The app is available for both Android and Apple devices. It is designated for use by authorized clinicians and is not intended for general use by patients.
The study, “Clinical Decision Support Tool and Rapid Point-of-Care Platform for Determining Disease Severity in Patients with COVID-19,” was published by Lab on a Chip.
Related Articles
New York Dentists Finally Return to Work as Challenges Remain
Dental School Developing Rapid, Low-Cost COVID-19 Test
DSO Turns Closed Offices Into COVID-19 Testing Sites












