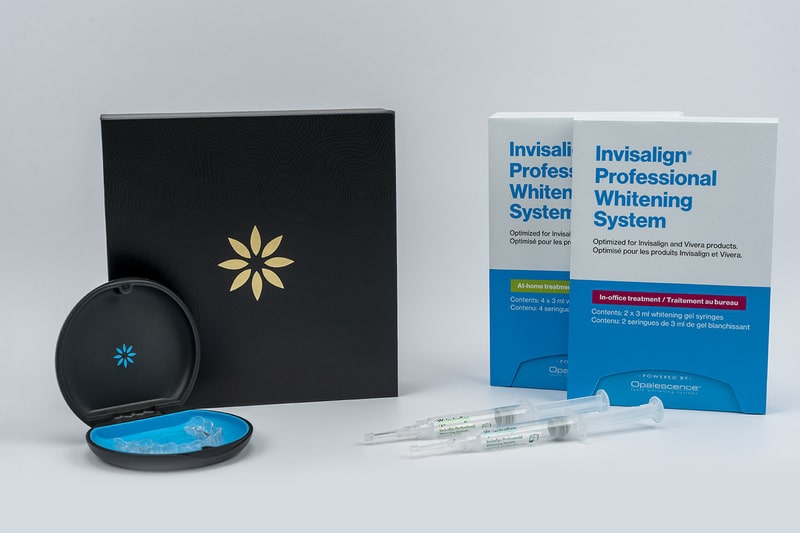
“Optimized for Invisalign Aligners and Vivera Retainers Powered by Ultradent’s Opalescence Tooth Whitening Systems”
Align Technology, Inc. (“Align”) (Nasdaq: ALGN) a leading global medical device company that designs, manufactures, and sells the Invisalign system of clear aligners, iTero intraoral scanners, and exocad CAD/CAM software for digital orthodontics and restorative dentistry, today announced an exclusive supply and distribution agreement with Ultradent Products Inc., a leading developer and manufacturer of high-tech dental materials, devices, and instruments worldwide.
As part of the multi-year agreement, Align will offer Invisalign trained doctors an exclusive professional whitening system with the leading Opalescence PF whitening formula from Ultradent, optimized for use with Invisalign clear aligners and Vivera retainers. The system will carry the co-branded name of ‘Invisalign Professional Whitening System – powered by Opalescence’ and will offer the same great whitening outcomes and streamlined practice experience dental professionals expect from the Opalescence PF product during active tooth movement with Invisalign aligners, as well as during passive retention using Vivera retainers. The Invisalign Professional Whitening System will be commercially available globally in 2022.
“A brighter, whiter smile is an important part of the Invisalign patient journey. In fact, a survey of North American Invisalign practices (1) shows that half of their patients ask for teeth whitening during or after they complete Invisalign treatment,” said Raj Pudipeddi, Align Technology chief product and marketing officer, and SVP and managing director of the Asia Pacific Region. “We believe that by providing an all-in-one solution that combines a leading teeth-whitening system with the most advanced clear aligner system in the world, we enable Invisalign trained doctors to enhance their patients’ treatment experience with a seamless workflow that also enables practice efficiency and growth. We’re very excited to partner with Ultradent to offer the first professional whitening system optimized for use with Invisalign clear aligners and Vivera retainers. Invisalign system trained doctors can use Opalescence PF for in-office teeth whitening treatment and for doctor supervised at-home whitening.”
“We’re honored to partner with Align to make the top professional teeth whitening products available to more clinicians and patients around the world,” said Ultradent President and CEO Dirk Jeffs. “Providing Align with teeth whitening products, ideal for use with Invisalign aligners and Vivera retainers, is a considerable step toward our mission of improving oral health globally and we look forward to pursuing this mission together.”
“Many of my patients would like whiter teeth as well as a straighter smile,” said Dr. Brian Amy, an orthodontist and Align Faculty member practicing in Oklahoma City. “The new Invisalign Professional Whitening System allows me to provide both simultaneously using the brands I trust.”
“After testing this system in my practice, I can confirm that the Invisalign Professional Whitening System is easy to administer and offers a great patient experience by combining whitening with Invisalign aligners or Vivera retainers,” said Dr. Jennifer Bell, a restorative and cosmetic dentist and Align GP Advisory Board Member practicing in Holly Springs, North Carolina. “The simple ordering mechanism minimizes burden on my practice to store and manage whitening products, while providing the brighter, whiter smiles we have come to expect from the Opalescence PF product.”
Ultradent is an ISO13485 certified facility and will manufacture the products according to those standards.
(1) 2017 online survey of Invisalign doctors, data on file at Align Technology.
About Align Technology, Inc.
Align Technology designs, manufactures and offers the Invisalign system, the most advanced clear aligner system in the world, iTero intraoral scanners and services, and exocad CAD/CAM software. These technology building blocks enable enhanced digital orthodontic and restorative workflows to improve patient outcomes and practice efficiencies for over 200 thousand doctor customers and is key to accessing Align’s 500 million consumer market opportunity worldwide. Align has helped doctors treat approximately 10.9 million patients with the Invisalign system and is driving the evolution in digital dentistry through the Align Digital Platform, our integrated suite of unique, proprietary technologies and services delivered as a seamless, end-to-end solution for patients and consumers, orthodontists and GP dentists, and lab/partners. Visit www.aligntech.com for more information.
For additional information about the Invisalign system or to find an Invisalign doctor in your area, please visit www.invisalign.com. For additional information about iTero digital scanning system, please visit www.itero.com. For additional information about exocad dental CAD/CAM offerings and a list of exocad reseller partners, please visit www.exocad.com.
About Ultradent Products, Inc.
Ultradent Products, Inc., is a leading developer and manufacturer of high-tech dental materials, devices, and instruments worldwide. Ultradent’s vision is to improve oral health globally. Ultradent also works to improve the quality of life and health of individuals through financial and charitable programs. For more information about Ultradent, call 800.552.5512, visit ultradent.com or find us on LinkedIn, Facebook, and Instagram.
Forward-Looking Statements
This news release contains forward-looking statements, including the expectations for and the terms of the agreement between Align and Ultradent the availability and capabilities of the combined Invisalign aligner and Opalescence offering, our beliefs regarding the market for the combined offering and the potential benefits to doctors, their offices and patients, and other similar comments or statements that are forward-looking in nature. Forward-looking statements contained in this news release relating to expectations about future events or results are based upon information available to Align as of the date hereof. Readers are cautioned that these forward-looking statements are only predictions and are subject to risks, uncertainties, and assumptions that are difficult to predict. As a result, actual results may differ materially and adversely from those expressed in any forward-looking statement.
Factors that might cause such a difference include, but are not limited to:
- the impact of the COVID-19 pandemic on the health and safety of our employees, customers, patients, and our suppliers, as well as the physical and economic impacts of the various recommendations, orders, and protocols issued by local and national governmental agencies in light of continual evolution of the pandemic, including any periodic reimplementation of preventative measures in various global locations;
- difficulties predicting customer and consumer purchasing behavior and changes in consumer spending habits as a result of, among other things, prevailing economic conditions, levels of employment, salaries and wages, and consumer confidence, particularly in light of the pandemic and as pandemic-related restrictions are eased regionally and globally;
- unexpected or rapid changes in the growth or decline of our domestic and/or international markets;
- increasing competition from existing and new competitors;
- rapidly evolving and groundbreaking advances that fundamentally alter the dental industry or the way new and existing customers market and provide products and services to consumers;
- the ability to protect our intellectual property rights;
- continued compliance with regulatory requirements;
- declines in, or the slowing of the growth of, sales of our intraoral scanners domestically and/or internationally and the impact either would have on the adoption of Invisalign products;
- the willingness and ability of our customers to maintain and/or increase product utilization in sufficient numbers;
- the possibility that the development and release of new products or enhancements to existing products do not proceed in accordance with the anticipated timeline or may themselves contain bugs or errors requiring remediation and that the market for the sale of these new or enhanced products may not develop as expected;
- a tougher consumer demand environment in China generally, especially for manufacturers and service providers whose headquarters or primarily operations are not based in China;
- the risks relating to our ability to sustain or increase profitability or revenue growth in future periods (or minimize declines) while controlling expenses;
- the impact of excess or constrained capacity at our manufacturing and treat operations facilities and pressure on our internal systems and personnel;
- the compromise of customer and/or patient data for any reason;
- the timing of case submissions from our doctors within a quarter as well as an increased manufacturing costs per case;
- foreign operational, political and other risks relating to our international manufacturing operations; and
- the loss of key personnel or work stoppages.
The foregoing and other risks are detailed from time to time in our periodic reports filed with the Securities and Exchange Commission, including, but not limited to, our Annual Report on Form 10-K for the year ended December 31, 2020, which was filed with the Securities and Exchange Commission (SEC) on February 26, 2021 and our latest Quarterly Report on Form 10-Q for the quarter ended June 30, 2021, which was filed with the SEC on August 4, 2021. Align undertakes no obligation to revise or update publicly any forward-looking statements for any reason.
RELATED ARTICLES
Align Technology To Showcase Digital Portfolio At The International Dental Show 2021
Align Technology Expands Global Operations to Support Growth
Align Technology Recognizes Teens for Their Community Efforts











