Free running (FR)1 pulsed Nd:YAG lasers have been used safely and effectively in the periodontal pocket for over 12 years for pocket sterilization2 and laser curettage.3 Laser periodontal therapy (LPT) is a laser-based procedure that builds on early laser sulcular debridement techniques, and was developed specifically for the treatment of moderate to severe periodontitis. It is a surgical procedure patterned after the excisional new attachment procedure (ENAP)4 to selectively remove diseased and necrotic tissue within the sulcus, but the laser has replaced the scalpel. Originally referred to as Laser-ENAP,5,6 LPT7,8 has evolved to provide a minimally invasive alternative to ressective, subtraction surgeries. Other advantages of LPT include improved hemostasis intraoperatively and improved patient comfort and acceptance. The procedure combines the best aspects of laser soft tissue surgery with well-established principles of periodontal disease therapy.
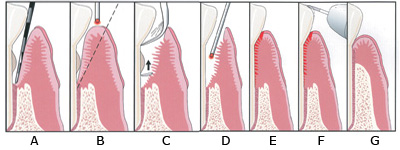 |
| Figure 1. Laser periodontal therapy, step-by-step technique. (A) Periodontal probing indicates excessive pocket depth. (B) Laser troughing: free running pulsed Nd:YAG laser irradiation, at 100 µsec to 150 µsec pulse duration. Beginning at the gingival crest (not into the sulcus at first). Troughing provides visualization of and access to the root surface by removing necrotic debris, releasing tension, and controlling bleeding. It further defines tissue margins preceding ultrasonic and mechanical instrumentation, preserves the integrity of the mucosa, and aids maintenance of the gingival crest. (C) A piezo-electric scaler, small curettes, and root files are used to remove root surface accretions. (D) A second pass with the laser at 150 µsec to 650 µsec pulse duration finishes debriding the pocket, provides hemostasis, and creates a “soft clot.” (E) The tissue is compressed against the root surface to close the pocket and stabilize the fibrin clot. (F) Occlusal trauma is adjusted with a high-speed handpiece, and mobile teeth are splinted. (G) New attachment (new bone, cementum, and PDL) is achieved. |
The cases presented here show that, for a growing number of patients, LPT can provide clinical outcomes comparable with invasive surgical procedures. The step-by-step surgical technique is outlined in Figure 1. This technique selectively removes sulcular and pocket epithelium, preserving connective fibrous tissues and Reté pegs.9 The primary end point of LPT is debridement of inflamed and infected connective tissue within the periodontal sulcus and removal of calcified plaque and calculus adherent to the root surface. In addition, the bacteriocidal effects of the FR pulsed Nd:YAG laser plus intraoperative use of topical antibiotics are designed for the reduction of microbiotic pathogens (antisepsis) within the periodontal sulcus and surrounding tissues.10-12 The wound is stabilized, the teeth are splinted, and occlusal trauma is minimized to promote healing. Oral hygiene is stressed, and continued periodontal maintenance is scheduled. No probing is performed for 6 months,minimum.
The desired result is achieving new attachment of gingival tissue to the root surface, thereby decreasing pocket depth. Ideally, remineralization and regeneration of supporting bone and regrowth of the periodontal ligament will also occur. The two cases presented were selected from our current database of patients, with complete probe depth and radiographic follow-up data.
CASE NO. 1
 |
| Figure 2. Preoperative x-ray. |
A 41-year-old white male presented August 23, 1994, with concerns regarding deep pockets around bridgework and a history of conventional scalpel gum surgery. The patient was referred by an existing patient of record and was under the care of a university periodontist. Medical history was unremarkable. Oral hygiene was fair. The condition of the gingiva was red and edematous. The lower left and right quadrants had bridgework to replace missing teeth, with several areas of malocclusion. Tooth No. 20 was vertically compressible in the pocket. The patient reported that in two previous scalpel surgeries the malocclusion was never addressed. Radiographs revealed moderate to severe horizontal and vertical bony defects throughout. Periodontal charting around 26 teeth (156 sites) demonstrated generalized moderate to deep pockets up to 12 mm deep; 104 sites probed ≥ 4 mm (36.5% normal) and 13 sites probed ≥ 8 mm. Mean probe depth (MPD) at baseline was 4.98 mm. A bilateral 12-mm defect was located on the distal-buccal and distal-lingual aspect of tooth No. 20 (Figure 2) with MPD of 9 mm. The patient was confirmed with a diagnosis of generalized moderate to severe periodontitis and was scheduled for LPT.
The upper left and lower right quadrants were treated on September 7, 1994. At that time we were using our original dLase 300, pulsed Nd:YAG dental laser (American Dental Technologies, Inc). In this system we had only one pulse width1 available, the 150 µsec cutting or “short pulse.”13 Troughing around each tooth was done at 150 mJ, 20 Hz (3 watts at the console with 3.5 watts measured at the fiber tip14,15). This laser did not have the capability of recording the total energy delivered, so the actual dosimetry is not available. A 320-µm optical fiber was used to deliver the laser energy. All quadrants were treated identically. The patient tolerated the procedure well with no complications or adverse effects.
The patient was evaluated 1 week following treatment of each quadrant. Soft tissue was healing well. Oral hygiene instructions and periodontal aids were dispensed. The patient was scheduled for a 60-day evaluation for periodontal probings and maintenance. When the patient was seen at 60 days the chart record states, “Periapical x-ray of No. 20 shows periodontal regeneration including PDL and bone. Oral Hygiene (OHI) is excellent. Bleeding on probing is minimal. Patient reports are excellent regarding the subjective and objective symptoms and signs. Patient is using Proxibrush regularly with excellent results.”
Mean probe depth change (MPDC) is computed by finding the mean of the differences in probing depths from baseline to follow-up. MPDC of tooth No. 20 at 14 months posttreatment reduced an average of 4.5 mm, or 50% pocket depth (PD) reduction with no recession. The 4.5-mm MPDC in this patient is consistent with a 4.5 MPDC for pockets ≥ 8 mm reported in a previous LPT case report of 4 pat
ients,7 and over 1,900 probing sites in 42 patients in a retrospective, multicenter, private practice study.8
 |
 |
| Figure 3. A 14-month postoperative x-ray. | Figure 4. Gamma correction of the before and 14-month postoperative x-ray using Emago software.16 White areas indicate increased bone density. Bone density increases extend circumferentially and three-dimensionally into the alveolus. |
 |
| Figure 5. A comparison of the pretreatment x-ray of the bone surrounding No. 24 with the x-ray obtained 9 and 14 months posttreatment shows strong evidence of substantial increased bone density. The density profiles at the bottom of Figure 5 demonstrate this clearly. Notice that the alveolar interproximal areas of teeth Nos. 23 through 25 are radiolucent before treatment, indicative of severe inflammation. By 14 months posttreatment, these areas have opacified. Radiographic findings show evidence of interproximal bone fill and what seem to be new bony structures interproximal to teeth Nos. 23 and 25. Notice that the densities in the region of the roots and the root canals have been balanced with gamma correction to yield approximate equal values. |
A comparison of the pretreatment x-ray of the bone surrounding tooth No. 20 with the x-ray obtained 14 months posttreatment shows strong evidence of bone fill mesially and distally (Figure 3). The gamma correction contrasts16 (Figures 4 and 5) demonstrate the elimination of the mesial and distal bony defect and a marked increase in radiographic density interproximal to teeth Nos. 19 through 21, throughout the alveolus and into bone. Mobility reduced from vertical class III to no mobility in 14 months. The results have been confirmed clinically and radiographically and have remained stable for 7.5 years with only one treatment.
CASE NO. 2
A 67-year-old white female presented with a large circumferential periradicular bone defect around tooth No. 24. She contacted our office after being referred to a local periodontist by her treating general dentist, upon the urging of her son who is a patient in our office.
The patient had an unremarkable medical history. Dental exam and history revealed a lower partial denture to replace the posterior teeth. The referring general dentist had, upon x-ray diagnosis of the defect on No. 24, immediately splinted the tooth with composite bonding to teeth Nos. 23 and 25. He had also taken the immediate step to adjust the incisal impact trauma.
All anterior teeth, especially teeth Nos. 8 and 9—those opposing teeth Nos. 23 and 24—were in fremitus upon immediate closing. The linguals of the anteriors were adjusted. Perio charting indicated the pockets and bone defects were on the lingual of the lower anteriors. Sinus/fistula tract was present on the lingual of tooth No. 24. Pretreatment, there was approximately 3 mm of recession on the lingual and 1 mm on the facial of tooth No. 24.
The PerioLase II (Millennium Dental Technologies) variable pulsed FR Nd:YAG laser was used for this patient. An improvement over the dLase 300 was the availability of seven user-selectable pulse durations (100 µsec to 650 µsec). For this case, troughing around the tooth was done with a “short pulse” having digital pulse duration of 100 µsec.13 Pulse energy was set to 150 mJ and repetition rate was 20 Hz, giving an average power of 3 watts.15 The parameters for the coagulating and hemostasis used to finish the procedure were 150 µsec duration, 150 mJ, 20 Hz, giving an average power of 3 watts.15 A 320-µm optical fiber was used to deliver the laser energy.
Another advantage of this laser system was the readout of total energy (in joules) delivered during the procedure, also known as fluence. This value is essential in determining the light dose.17 To compute light dose, the total energy delivered (100 J) is divided by the sum of the depths of all pockets (56 mm), which, in this case, was 1.78 J per mm PD. (Please note: The proper light dose used in this case is counter-intuitive for a defect as large as this. The total amount of energy used—1.78 J/mm PD is about 10 times less than the 16 J/mm PD used for most mean pocket depths reported previously.7,14,15)
 |
 |
| Figure 6. Mesial of tooth No. 24 at 9 months post-op. | Figure 7. Distal of tooth No. 24 at 9 months post-op. |
 |
| Figure 8. Lingual of tooth No. 24 at 9 months post-op. |
The patient tolerated the procedure well, with no complications or adverse effects. No dressings were placed immediately post-op to protect the gum tissues, only the stable fibrin “soft” clot that was forming. The condition of the periodontal tissues 1 week post-op was remarkable in the appearance of healing in spite of the large size of the defect. No evidence of further recession was noted. Periodontal maintenance by the patient was dutiful and consistent at 3-month intervals. A follow-up exam was done at 9 months posttreatment; 1 mm of recession on the facial was noted (Figures 6 and 7), and 3 mm of recession on the lingual (Figure 8), along with light calculus and a healthy appearing gingiva.
Probing depths charted 9 months posttreatment provided a quantitative index of the efficacy of LPT. The total MPD for tooth No. 25 was 9.3 mm pretreatment, and 2.8 mm at 9 months, for a MPDC of 69.89% pocket reduction or 6.5 mm overall. The pockets ≥ 8 mm have been reduced 90.6% by an average of 7.6 mm from MPD of 10.6 mm pretreatment, to an MPD of 3 mm. The MPDC in this case study of 6.5 mm and 7.6 mm exceeds the 4.5 mm reported in case No. 1 and in previous reports. Note: This patient was treated only once during the entire 14-month time frame. Periodontal probin
gs were not performed until 9 months post-op.
DISCUSSION
More detailed, quantitative comparative analyses of the efficacy of LPT with a larger sample size are in preparation. Although presented as unique examples, these cases can be compared with published data for a better understanding of how these patients have fared relative to alternative treatments.
In more than 24 combined years of research and clinical laser experience, we have had the opportunity to use most laser wavelengths, device configurations, and delivery systems. We have applied our experience and research to calibrate the laser parameters and modify our protocol as we went along. When we evolved from using the dLase 300 to the Multi-Variable Pulse PerioLase II, the addition of longer pulse durations dramatically improved intraoperative hemostasis and shortened the overall time in the chair by 50%, from 90 minutes per quadrant to 45 minutes. The energy readout (fluence) has allowed us to keep track of light dose and to compare dosimetry with clinical outcomes. From this we have developed procedural-based dosimetries. We have also added modifications to the overall treatment program. Over the years, LPT has evolved to the proper application of laser technology plus a medically sound approach to wound management, together with real-world clinical efficiencies in implementation and applicability.
The changes we have observed in bone density are very similar to those reported by Dubrez et al.18 Comparison of our preliminary radiographic findings with this study are useful because their quantitative radiographic analyses are identical in concept to the density profiles generated by Emago. In the Dubrez et al18 study, interproximal sites in patients who demonstrated bone loss in association with ≥ 5-mm PDs were evaluated. Treatment consisted of coronal scaling, root planing, topical antibiotics, and rigidly maintained oral hygiene. They observed improved bone density at 1 year. A quantitative comparison of bone density changes seen in that study versus what we observe in our patients is beyond the scope of these case reports. However, it is not unexpected that if we see clinically a general improvement in the health of the surrounding tissues we will also see a subsequent increase in the radiographic density of alveolar bone.
CONCLUSION
It is our hope that the case studies presented here demonstrate what is possible to achieve if the complete disease processes are understood and addressed. While it is possible to achieve results similar to those shown in this article without using a pulsed Nd:YAG fiber-optic laser, these results were accomplished using a “closed” noninvasive methodology that was both easy to perform and perceived by the patients as a much preferred option to scalpels, bone grafts, or extraction and implant placement.
Acknowledgments
The authors thank David M. Harris, PhD, of Bio-Medical Consultants, Inc, in Castro Valley, California, for data processing and graphic presentations, and Alan Rosenberg, DDS, for his pretreatment anterior splinting and incisal adjustments.
References
1. Free Running is a description of a laser’s temporal emission mode (TEM). Free running pulsed lasers have pulse durations (or pulse times) measured in millionths of a second (10-6 seconds)—”microsecond” and abbreviated with a “µ” preceding “sec.” Pulse duration (aka pulse width) refers to the length of time a given laser pulse is “on” before shutting off and allowing for thermal relaxation (cooling) of the target tissue. Free running pulsed lasers allow for high peak powers in the thousands of watts and extremely long “off” times (eg, 500 x’s) which allows for average powers of 3 to 4 watts.
2.McCarthy DK. Laser Curettage Using the Pulsed Nd:YAG Laser in Vivo. North American Academy of Laser Dentistry: Boston, Mass; 1990.
3. Midda M. Nd:YAG Subgingival Curettage. Innovation et technologie en biologie et medicine. Actes du deuxienne congre modial. L, impact des lasers en sciences odontologiques. Paris, France: [Presentation] 1990:105.
4. Yukna RA, Bowers GM, Lawrence JJ, et al. A clinical study of healing in humans following the excisional new attachment procedure. J Periodontol. 1976;47:696-700.
5. Gregg RH, McCarthy DK. Laser ENAP for periodontal bone regeneration. Dent Today. 1998;17(5):88-91.
6. Gregg RH, McCarthy DK. Laser ENAP for periodontal ligament regeneration. Dent Today. 1998;17(11):86-89.
7. Gregg RH, McCarthy DK. Laser periodontal therapy: case reports. Dent Today. 2001;20(10):74-81.
8. Harris DM, Gregg RH, McCarthy DK, et al. Sulcular debridement with pulsed Nd: YAG. In: Lasers in Dentistry. (4610-37). Bellingham, Wash: SPIE; 2002. In Press.
9. Gold SI, Vilardi MA. Pulsed laser beam effects on gingiva. J Clin Periodontol. 1994;21:391-396.
10. Midda M, Renton-Harper P. Lasers in dentistry. Sr Dent J. 1991;170:343-346.
11. Moritz A, Schoop U, Goharkhay K, et. al. The bactericidal effect of Nd:YAG, Ho:YAG and Er:YAG laser irradiation in the root canal: an in vitro comparison. J Clin Laser Med Surg. 1999;17:161-164.
12. Whitters CJ, MacFarlane TW, McKenzie D, et. al. The bactericidal activity of pulsed-Nd:YAG laser radiation in vitro. Lasers in Medical Science. 1994;9:297-303.
13. Pulse Duration can be measured several ways depending on whether the pulse is digital or analog. Digital pulse durations are qualitatively and quantitatively different than analog pulse durations. An analog pulse has a Gausian profile (ie, a sine wave), where the digital pulse is square. Digital pulse durations are more accurately measured than analog because the shape of the area measured is a discrete area versus an alternating wave front. The convention used here is known as full width/half max. That is the pulse time (duration) in microseconds measured the full width on the “x” axis (width) of an oscilloscope at one half the maximum of the “y” axis.
14. CAUTION: Laser dosimetry described in this paper is NOT recommended unless the practitioner is well-trained and experienced. Exceeding the laser parameters or overtreating large defects described for these cases may lead to prolonged healing, tissue and tooth loss, and other complications.
15. Power (Watts): The rate of doing work. It is critical to accurate communications of dosimetry that therapeutic power delivered to tissue be confirmed through measurement at the fiber tip with a calibrated power meter, as the power can vary as much as 30% or more from the power settings displayed on the console of any laser device. A PowerMax PM600 power meter (Molectron Detector) was used in both case studies presented.
16. Analog x-ray films were digitized and analyzed with Emago (Advanced Medical Devices) and Photoshop software. In Emago the pretreatment reference image and the posttreatment images were balanced for differences in exposure and film processing characteristics with gamma correction. Gamma correction is a computerized algorithm that modifies the gray level distribution of an image using the gray level distribution of another image as a reference. The software provides means of quantitative analysis of corrected radiographic densities through the use of density profiles. The density profiles shown in Figures 4 and 5 represent a plot of the gray value of each pixel along a line through the x-ray that is selected by the user. Each gray value represents the relative radiographic density at that point in arbitrary units. Emago geometric reconstruction is a useful alternative to obtaining before and after x-rays with identical projection geometry. In subtraction radiograp
hy this is provided by consistently using individual bite blocks or other aiming techniques (refs). Emago geometric reconstruction produces a pair of images with identical image formation geometry by mapping the information contained in one image onto the projection plane of a reference image. After projection corrections and density analysis, the two images were combined to produce one side-by-side image that was filtered in Photo Shop (contrast and sharpness) to enhance visualization of bony features.
17. Light dose (joules per mm pocket depth) is similar to drug dose (mg per kg body weight) in that light dose defines the concentration of laser energy at the treatment site in a similar manner as drug dose defines the concentration of a drug in the tissues. Light dose is a very useful parameter inasmuch as certain clinical outcomes of laser surgery (eg, adverse effects) are dose dependant.
18. Dubrez B, Graf JM, Vuagnat P, Cimasoni G. Increase of interproximal bone density after subgingival instrumentation: a quantitative radiographic study. J Periodontol. 1990;61(12):725-731.
Dr. Gregg is a past faculty member at UCLA School of Dentistry, Section of Hospital Dentistry. He has been using lasers clinically since August 1990, including CO2, pulsed Nd:YAG, surgical and photopolymerization argon, and Er:YAG. He has given lectures nationally and internationally on the subject of clinical laser applications, and has conducted seminars for UCLA Department of Continuing Education. In addition to authoring several peer-reviewed articles on the clinical applications of pulsed Nd:YAG for endodontic and periodontal uses, he is an author of the Laser Curriculum Guidelines, versions 1 and 2. Dr. Gregg has obtained his Mastership and Educator’s Certification in the Academy of Laser Dentistry. He may be contacted at (562) 860-6587 or [email protected].
Dr. McCarthy is a past faculty member at UCLA School of Dentistry, Section of Advanced Education General Dentistry (AEGD). He has been using lasers clinically since November 1989, including CO2, pulsed Nd:YAG, surgical and photopolymerization argon, and Er:YAG. He has given lectures nationally and internationally on the subject of clinical laser applications, and has conducted seminars for UCLA Department of Continuing Education. In addition to authoring several peer-reviewed articles on the clinical applications of pulsed Nd:YAG for periodontal uses, he is an author of the Laser Curriculum Guidelines, versions 1 and 2. Dr. McCarthy served as an executive officer in the International Academy of Laser Dentistry and wrote its constitution and by-laws. He has obtained his Mastership and Educator’s Certification in the Academy of Laser Dentistry. He may be reached at (562) 860-2908 or [email protected].
Disclosure: Drs. Gregg and McCarthy developed the FDA-cleared PerioLase pulsed Nd:YAG laser, and are founders of Millennium Dental Technologies, Inc. They developed and hold the patent for the Laser ENAP periodontal technique.


