AESTHETIC DILEMMA OF THE GINGIVAL MARGIN
The data is clear: Porcelain and composite resin are both biocompatible at the gingival margin and well-tolerated.1-3 One major cause for concern is that composite can cause tissue irritation if it impinges directly on the gingiva.4-6 This may ultimately cause rather unaesthetic gingival recession. Porcelain is less challenging, but marginal plaque buildup, often stimulated by the thin layer of exposed resin cement, can lead to a similar irritation of the free gingiva and, ultimately, recession.7,8
Even skillful subgingival margin placement will typically, within 3 to 5 years, whether due to aging, gingival irritation, or inadequate home maintenance by the patient, lead to unaesthetic recession and a highly visible exposure of the darker dentinal surface.9,10 The best restorative approach is to position composite margins supragingivally,3,11 with the resin material reasonably away (0.25 to 0.5 mm) from the soft periodontal structures, decreasing the likelihood of tissue irritation.
Today’s adhesives and restoratives make this a rather straightforward task.12-19 Later-generation adhesives bond predictably and relatively equally to both enamel and dentin.14,15 Generally, seventh- and eighth-generation adhesives exhibit similar attachment to both enamel and dentin, eradicating the stresses that would be caused by unequal polymerization contraction.17-19 It is therefore clinically realistic to place a restoration that is continuous through the dentino-enamel junction (DEJ), covering as much enamel and dentin as is required due to decay, abfraction, or aesthetics.
A significant aesthetic predicament quickly presents, however. Most dental restorative materials are standardized to the enamel shades. While some “dentin” shades are available, they often do not closely approximate the darker coloration of exposed dentin, particularly with endodontically treated teeth.
When restoring a Class V decay, abfraction, or a small gingival recession, an enamel-shaded resin is bonded at the labial DEJ. A typical central incisor is about 10.5 mm cervico-incisally (CI).20 Any significant increase in the apparent length of a tooth, specifically in the anterior labial region, will undermine the aesthetics of the smile. Increasing the maxillary central incisor’s CI aspect by as little as 3 mm (Figure 1) adds 30% to the apparent vertical dimension, significantly altering the CI:MD ratio. This upsets the aesthetic parameters of the smile (Figure 2).
For patients with moderate recession, the visual discrepancy is further reinforced. The CI:MD ratio impact is more dramatic with maxillary laterals and mandibular incisors (with an average of 9.0 mm CI). Maxillary and mandibular cuspids (clinically longer at 10 to 11 mm CI) are often the teeth most prone to gingival recession and also the most visible, both anteriorly and laterally.
Patients with gingival recession, abfraction, and/or decay tend to look older than their age. Unfortunately, hiding the darker root dentin with an enamel-colored resin only serves to make these teeth more noticeable; seemingly longer; and, consequently, less appealing.
The practitioner must solve these problems practically, aesthetically, and with minimum invasiveness. The ideal treatment is functional, restoring missing enamel and dentin to natural dimensions and contours. Lost enamel is replaced with enamel-shaded composite resin. Receded gingiva is replaced with gingival-shaded composite resin. Their interface is an artificial enamel-gingival junction in composite, restoring the aesthetics of the patient’s smile.
SOLVING THE GINGIVAL AESTHETIC DILEMMA
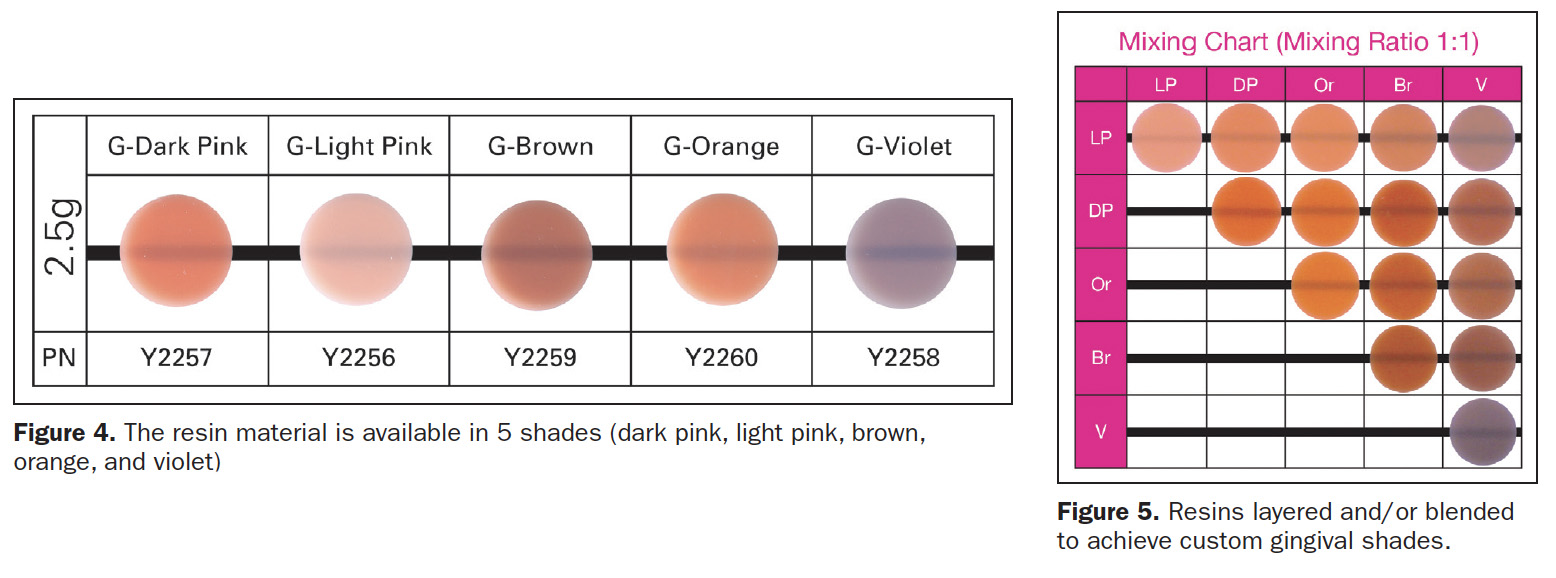
Beautifil II Gingiva (Shofu Dental) was developed to rebalance the pink aesthetics of the dental cervical areas (Figure 3). Beautifil II Gingiva’s indications for use include wedge-shaped defects, cervical decay, the aesthetic rectification of gingival recession, shielding exposed cervical areas, and splinting of mobile teeth. Gingiva resin is available in 5 hues (dark pink, light pink, brown, orange, and violet) (Figure 4) that may be layered and/or blended to create custom shades for various hues of gingival pigmentation according to specific clinical needs (Figure 5).
Beautifil II composite resin is highly aesthetic, fluoride-releasing, and indicated for all classes of restorations. Many published studies over the past 20 years have demonstrated no failures, no secondary caries, no postoperative sensitivity, and high retention of both color stability and surface luster. Its chemistry is founded on Shofu’s proprietary Giomer technology (Figure 6). Giomer resins have a significant advantage: They release fluoride, protecting dental structures at the restorative margin, and their fluoride content is rechargeable by toothpastes, fluoride rinses, and varnishes.
Hence, Giomer fluoride releasing capacity does not decrease over time.
It is essential that the restorative gingival margin (whether pink or enamel in color) is positioned supragingivally and somewhat away from the free gingival margin.3,4,6 While a very narrow darker band of dentinal structure may be visible apically, the restoration’s coronal enamel and pink gingival coloration focus attention away from this region. Gingivally blended restorations allow the professional to deliver both aesthetic and supragingival margins in the same restoration. The supragingival margin also facilitates effective home maintenance for the patient.
Restorations that are very close to the free gingival margin require effective moisture and bleeding control. Rubber dam techniques are not practical in the apical working area. Retraction cords may physically or chemically compromise the restorative materials. For the conscientious patient, good oral hygiene produces a healthy gingival microenvironment: minimal pocketing and no bleeding on probing. For most patients, however, the practitioner must revise the gingival state to increase the likelihood of clinical success. The most predictable technique for tissue sculpting is the diode laser.21-23 Utilizing low power (1 to 1.5 W), an ideal, dry, clean, and blood-free working area can be achieved in less than a minute.
CASE REPORTS
Case 1: Visible Recession and Decay
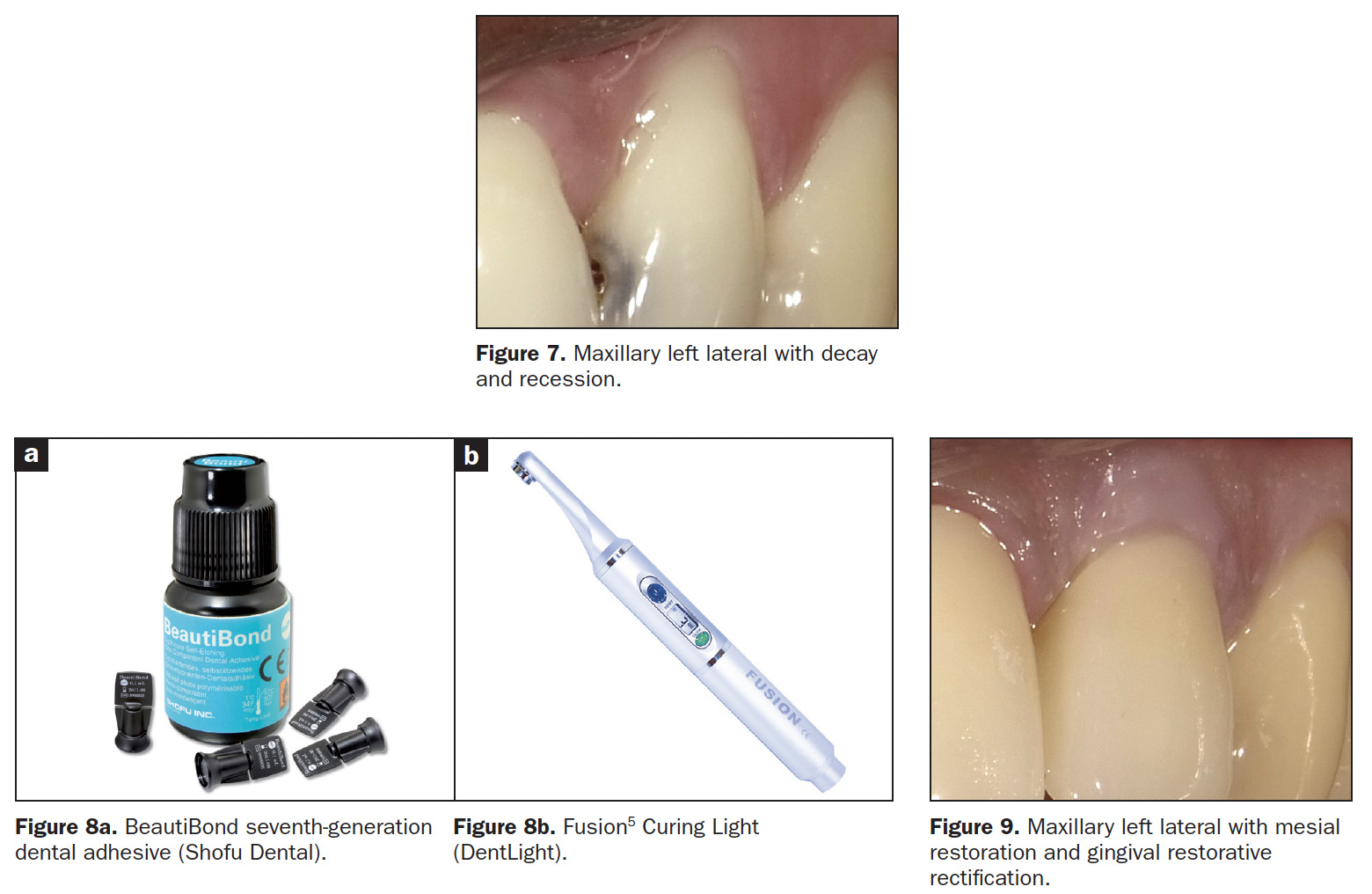
The patient’s main complaint was the gingival recession on the left maxillary lateral, not the mesial caries (Figure 7). As the patient’s home care was effective, the MLB restoration was straightforward using BeautiBond (Figure 8a) and Beautifil Flow Plus X, both from Shofu Dental. The steps for the gingival aesthetic restoration of the buccal recession are:
- Gently micro-abrade the receded area and the apical enamel, removing food debris and plaque. The abrader nozzle must be inclined incisally to prevent gingival irritation and bleeding. Rinse thoroughly and lightly air dry. The surface is left slightly moist (moistness level is not critical).
- Apply BeautiBond, a seventh-generation adhesive, and leave for 10 seconds. Thoroughly air dry the adhesive (very critical). Light cure utilizing the Fusion5 Curing Light (DentLight), offering deep cure of composite resins within 3 seconds with a uniform 4,000 mW/cm2 output (Figure 8b).
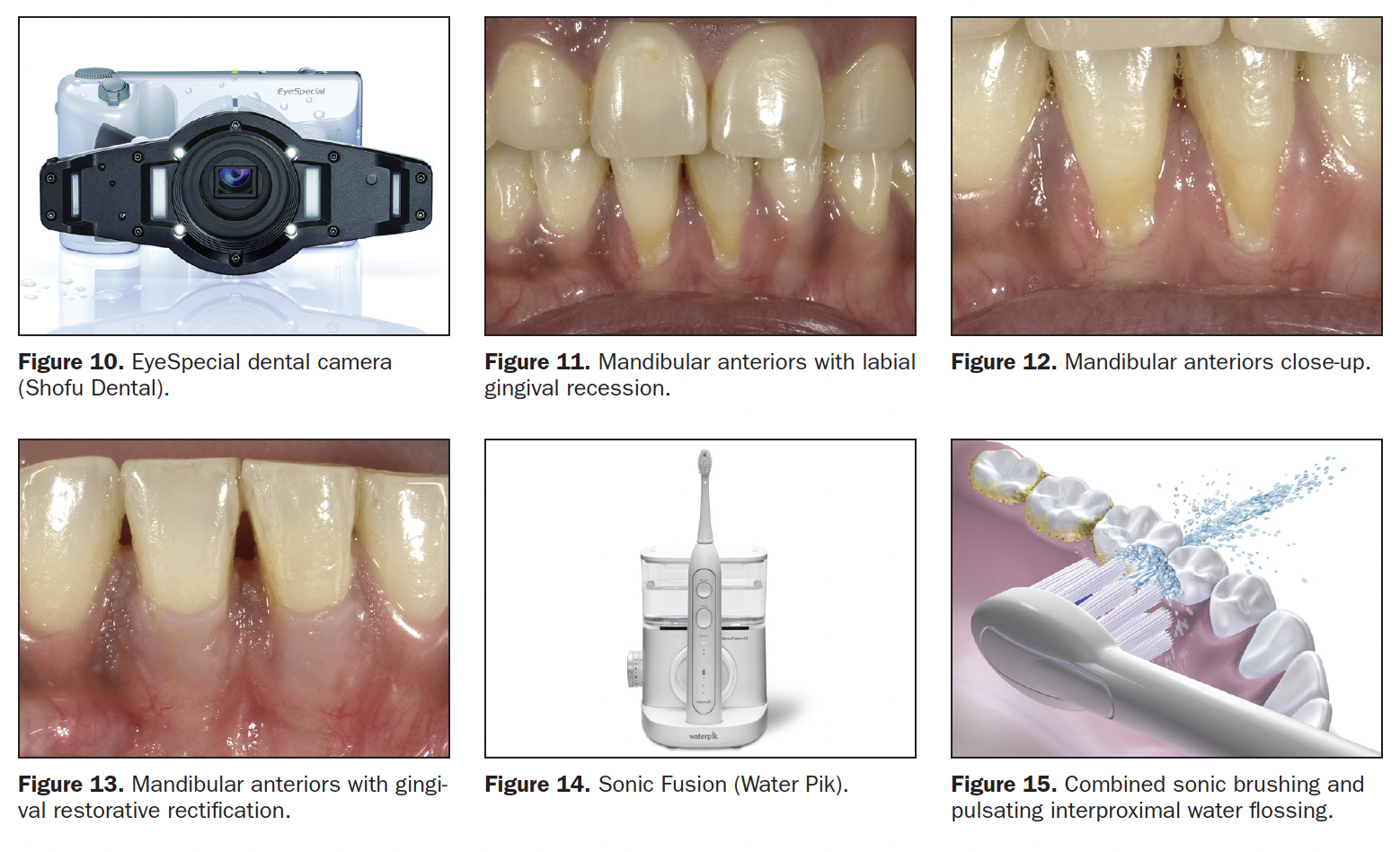
- Select the Beautifil II Gingiva shade (Light Pink and Violet for this patient) and place it in the receded area, restoring the buccal dimensions and contours of the original soft tissue, then light cure. Leave a 0.25- to 0.5-mm gap between the apical restorative margin and the free gingival margin (Figure 9). This spacing will avoid gingival irritation; is easily maintainable by the patient; and is not readily visible, even in close-up photographs. (All photography was done with the EyeSpecial camera [Shofu Dental] [Figure 10]).
Case 2: Mandibular Anteriors Recession
This recession is encountered very often. Lower anteriors are small, closely positioned, and not easily cleansable by tongue and lower lip (Figure 11). Gravity and the upright position cause food debris and plaque to accumulate labially and interproximally (Figure 12). For this patient, the likely cause for the recession was the frenum that was attached to the apical gingiva. The preliminary treatment was a diode laser frenectomy to eliminate the muscular pull.21-23 The steps for the gingival aesthetic restoration of the buccal recession are:
- Gently micro-abrade (in the incisal direction) the receded areas and the enamel nearest to the DEJ, removing food debris and plaque. Rinse thoroughly and lightly air dry. The surface is left slightly moist.
- Apply BeautiBond, and leave it for 10 seconds. Thoroughly air dry the adhesive, then light cure.
- Select the appropriate Beautifil II Gingiva shade (Light Pink) and apply to the receded area to restore the buccal dimension and contour of the original soft tissue, then light cure. Leave a 0.25- to 0.5-mm gap between the apical restorative margin and the free gingival margin (Figure 13). This procedure restores the expected vertical height of the unreceded gingiva, making the patient’s smile look more aesthetic and younger.
Effective, long-term home maintenance must include procedures that are familiar to the patient and easy to implement. Toothbrushing is effective and in widespread use. Interdental string flossing is less so. While string flossing is well-documented and encouraged, patient resistance to the process and inconsistent home use limit its benefits. The Water Flosser (Water Pik) facilitates the interdental cleaning process and has been shown to improve the results.24 Many models are available, including the Sonic Fusion (Water Pik), which offers simultaneous water flossing and sonic brushing (Figures 14 and 15).
CONCLUSION
Beautifil II Gingiva offers the practitioner the solution to the gingival aesthetic dilemma. The aesthetic restoration of the gingiva is predictable and is often completed without local anesthetic or patient discomfort. These restorations are fully functional and restore both hard and soft dental structures to natural dimensions and contours with minimum invasiveness. The restoration of the coronal anatomy with tooth-colored composite resins is well-documented and quite routine; the development of the artificial enamel-gingival junction and the technique for reconfiguring missing gingival structures with composite resins is a novel solution that restores the patient’s smile and facial aesthetics.
REFERENCES
1. Freedman G. Ultraconservative porcelain veneers. Esthet Dent Update. 1997;2:224-228.
2. Freedman G. Ultraconservative rehabilitation. Esthet Dent Update. 1991;5:80-85.
3. Freedman G, Fugazzotto PA, Greggs TR. Aesthetic supragingival margins. Pract Periodontics Aesthet Dent. 1990;2:35-38.
4. Newcomb GM. The relationship between the location of subgingival crown margins and gingival inflammation. J Periodontol. 1947;45:151.
5. Renggli H, Regolati B. Gingival inflammation and plaque accumulation by well adapted supragingival and subgingival proximal restorations. Helv Odont Acta. 1972;L6:99.
6. Waerhau SJ. Histologic considerations which govern where the margins of restorations should be located in relation to the gingiva. Dent Clin North Am. 1960;4:161.
7. Berman M. The complete coverage restoration and the gingival sulcus. J Prosthet Dent. 1973;29:1301.
8. Marcum O. The effect of crown margin depth upon gingival tissues. J Prosthet Dent. 1967;17:479.
9. Freedman G, McLaughlin G. The Color Atlas of Porcelain Laminate Veneers. Ishiyaku EuroAmerica, Inc.; 1990.
10. Freedman G. Chapter 23: Cements. In: Contemporary Esthetic Dentistry. Elsevier Publishing; 2011:551.
11. Freedman G, Klaiman HF, Serota KT, et al. EndoEsthetics: Part II. Castable ceramic post and core restorations. Ont Dent. 1993;70:21-24.
12. Albers HF. Dentin-resin bonding. Adept Report. 1990;1:33-34.
13. Munksgaard EC, Asmussen E. Dentin-polymer bond promoted by Gluma and various resins. J Dent Res. 1985;64:1409-1411.
14. Barkmeier WW, Erickson RL. Shear bond strength of composite to enamel and dentin using Scotchbond multi-purpose. Am J Dent. 1994;7:175-179.
15. Swift EJ, Triolo PT. Bond strengths of Scotchbond multi-purpose to moist dentin and enamel. Am J Dent. 1992;5:318-320.
16. Gwinnett AJ. Moist versus dry dentin; its effect on shear bond strength. Am J Dent. 1992;5:127129.
17. Freedman G, Leinfelder K. Seventh generation adhesive systems. Dentistry. 2003; 1:15–18.
18. Freedman G. 7th generation adhesive systems. Dental Asia (English). 2019; 2:50-53.
19. Freedman G. Adhesion: past, present, and future. Oral Health. 2019;109:7-14,82.
20. Wheeler RC. A Textbook of Dental Anatomy and Physiology. W.B. Saunders; 1965.
21. Goldstep F. Soft tissue diode laser: where have you been all my life? Oral Health. 2009;99:7;34-38.
22. Goldstep F. Diode lasers for periodontal treatment: the story so far. Oral Health. 2009;99:12,44-46.
23. Goldstep F. Diode lasers: the soft tissue handpiece. Dental Asia (English). 2011;1:28-32.
24. Rosema NA, Hennequin-Hoenderdos NL, Berchier CE, et al. The effect of different interdental cleaning devices on gingival bleeding. J Int Acad Periodontol. 2011;13(1):2-10.
ABOUT THE AUTHORS
Dr. Freedman is co-founder and past president of the American Academy of Cosmetic Dentistry, co-founder of the Canadian Academy for Esthetic Dentistry, regent and Fellow of the International Academy for Dental Facial Esthetics (IADFE), and Diplomate and chair of the American Board of Aesthetic Dentistry. He is an adjunct professor of dental medicine at Western University in Pomona, Calif, and professor and program director of the MClinDent Programme in Restorative and Cosmetic Dentistry at BPP University in London. Dr. Freedman is the author of 14 textbooks, more than 900 dental articles, and numerous webinars. He can be reached at freedman@epdot.com.
Disclosure: Dr. Freedman consults for more than 50 companies.
Dr. Lalla is a graduate of the University of The West Indies and practices dentistry in Trinidad and Tobago with a special focus on cosmetic and implant dentistry. Following an AEGD from Lutheran Medical Center, he has been accorded Fellowships in the International Congress of Oral Implantology, the American Academy of Implant Prosthodontics, and the IADFE. He can be reached at tropicaldental@yahoo.com.
Disclosure: Dr. Lalla reports no dislcosures.
RELATED ARTICLES
Nanocomposites Provide Reliability and Aesthetics
Options in Restorative Dentistry: Resin-Modified Glass Ionomers
Creating Precision Restorations Using a Hand-Dispensed Polyether












